CASE REPORT
Infective Endocarditis Related to Intravenous Drug User: Report of Four Cases
Ermira Muço1, *, Arjan Harxhi1, Amela Hasa2, Pëllumb Pipero1, Arta Kushi1, Irena Ceko3, Edmond Puca1, Dhimitër Kraja1
Article Information
Identifiers and Pagination:
Year: 2018Volume: 10
First Page: 1
Last Page: 8
Publisher ID: TOIDJ-10-1
DOI: 10.2174/1874279301810010001
Article History:
Received Date: 02/11/2017Revision Received Date: 22/01/2018
Acceptance Date: 07/03/2018
Electronic publication date: 14/03/2018
Collection year: 2018

open-access license: This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International Public License (CC-BY 4.0), a copy of which is available at: https://creativecommons.org/licenses/by/4.0/legalcode. This license permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objective:
To describe the clinical, laboratory, microbiological, and echocardiographic findings in four intravenous drug users with endocarditis hospitalized and followed in our Infectious Disease Service, a tertiary university hospital as well as to determine the efficacy of medical treatment.
Methods:
From a database of 35 subjects with endocarditis during five years, we made a retrospective analysis of data for four cases between the age of 24-33 years old which were intravenous drug users.
Results:
Infective endocarditis was encountered in four drug users with positive blood cultures (Staphylococcus aureus was present in all the cases), vegetations in the tricuspid native valve in ultrasound, high fever (more than 38oC). The four cases were male and the mean age was 29 years (range 24-33 years). Three out of the four cases presented with pulmonary involvement and only one with femoral and popliteal vein thrombosis. Two out of four cases had acute renal and hepatic failure and only one had acute cutaneous vasculitis. Transesophageal Echocardiography (TEE) was also performed in two cases. For all of them medical management consisted of antibiotic therapy and two out of them underwent surgery because of the persistence of valvular vegetations after antibiotic therapy. The prognosis was good with 0% mortality.
Conclusion:
Infective endocarditis should be considered in the differential diagnosis of intravenous drug users presenting with various clinical scenarios. Echocardiography remains the main modality and should be used serially to facilitate early diagnosis. The successful management of a complicated case often requires the close cooperation of an infectious disease physician, a cardiologist, an addiction physician and occasionally a cardiac surgeon.
1. INTRODUCTION
Endocarditis, also called infective endocarditis, is an infection and inflammation of the heart valves and the inner lining of the heart chambers, which is called the endocardium. The infecting organism enters the bloodstream through a break in the skin caused by a skin disorder or injury; a medical or dental procedure; or a skin prick, especially among intravenous drug users. Infective Endocarditis (IE) is one of the most severe complications in intravenous drug abusers. The tricuspid valve is the most frequently affected [1]. Staphylococcus aureus is the most common etiologic agent [2]. It is also estimated that the incidence of IE in intravenous drug abusers is 2 to 5% per year and the overall death rate is 5 to 10% [3].
2. METHODS
The material for this study is obtained from records of patients maintained by the authors in a tertiary care teaching institute. From a database of 35 subjects with endocarditis, we studied four cases between age of 24-33 years old which were intravenous drug users. The records of these cases form the material for this analysis. Diagnosis of Infectious Endocarditis was based on the modified Duke criteria [4]. We studied parameters as patient personal data (age, sex, educational level), risk factors, causative microorganism, number of positive blood cultures, duration of symptoms, type of valve involved, echocardiographic findings (vegetation site, vegetation size, complications); other radiological examinations results (electrocardiogram, chest radiograph, chest CT-scan); presence and type of embolic events; treatment and mortality. Detailed laboratory workup including complete blood count, Erythrocyte Sedimentation Rate (ESR), renal and liver function test and blood cultures were studied (Table 1).
| Epidemiological, Clinical, Laboratory, Radiological Data | Case 1 | Case 2 | Case 3 | Case 4 |
|---|---|---|---|---|
| Patient´s Age | 33 years old | 24 years old | 30 years old | 29 years old |
| Gender | Male | Male | Male | Male |
| Duration and Kind of Drug Consumption | 11 years /Heroine | 5 years/Heroine | 9 years/Heroine | 7 years/Heroine |
| Duration of Symptoms | 5 days | 18 days | 23 days | 6 days |
| Symptoms | cough, chest pain, fatigue, fever, myalgia, arthralgia. | cough, chest pain, dyspnea, fever, myalgia, arthralgia,. | cough, chest pain, fatigue, high fever, myalgia, arthralgia | high fever, myalgia, arthralgia, fatigue |
| Etiological Agent | Staph. aureus | Staph. Aureus | Staph. aureus | Staph. Aureus, Acinetobacilis baumani, Strept.pneumonie |
| HIV | negative | Negative | Negative | Negative |
| Valve and Type of Valve Involved, Number and Size Vegetations. | Tricuspid, native, 1 vegetation, 2.7cm2 | Tricuspid, native, 1 vegetation, 2.87cm2 | Tricuspid, native, multiple, 2-2.5 cm2 | Tricuspid, native, 1 vegetation, 0.4 cm2 |
| Vascular Phenomena | Septic pulmonary emboli | Septic pulmonary emboli | Septic pulmonary emboli, cutaneous vasculitis | |
| Events Observed | Staphylococcic pneumonia, acute renal and hepatic failure, conjunctival hemorrhage |
Staphylococcic pneumonia, Popliteal-femoral venous thrombosis | Staphylococcic pneumonia, acute cardiac, renal and hepatic failure, cutaneous vasculitis | Popliteal-femoral venous thrombosis |
| Comorbidity | No | Hepatitis C | Hepatitis B, Hepatitis C | Hepatitis C |
| Surgery | No | Yes | No | No |
| Death | No | No | No | No |
| Duration of Hosp. | 54 days | 43 days | 63 days | 23 days |
| Treatment | Vancomycin2g/day, Gentamycin 1 mg/kg every eight hours, Cefepime4g/day; | Vancomycin 2g/day, Merepenem3g/day | Vancomycin2g/day, Merepenem3g/day, Rifampicine 600mg/day; | Vancomycin2g/day, Gentamycin1 mg/kg every eight hours, Ceftriaxone 2g/day |
3. RESULTS
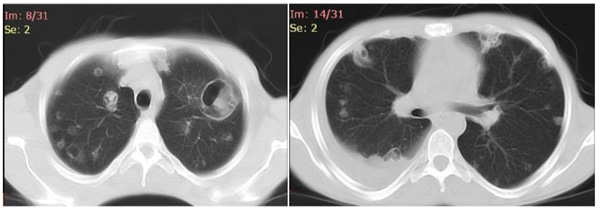
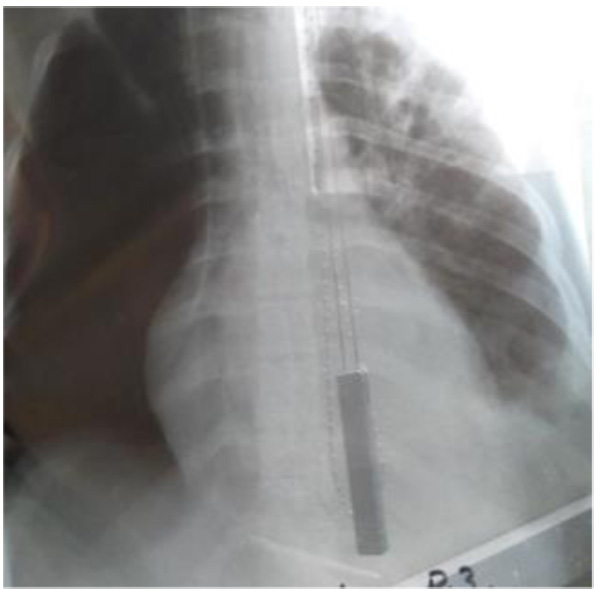
All the four patients were men with a mean age of 29 years (range 24-33 years), and drug users. During hospitalization, none of the patients developed physical withdrawal state from heroin. Three patients during their hospital stay have been in Methadone maintenance treatment. In all the cases we diagnosed right-sided endocarditis with tricuspid valve affection. Echocardiography identified vegetations (1 to multiple in case 3) that ranged in size from 2cm2 to 2.8cm2 (Figs. 1 and 2). The first three cases had positive blood cultures for Staphylococcus aureus while the fourth case besides Staphylococcus aureus had also Acinetobacilis baumani and Streptococcus pneumonie. Fever was presented in all the cases. In cases 1, 2, 3, complications attributed to septic pulmonary emboli are pulmonary abscesses, unilateral pneumothorax, bilateral pleural effusions and empyema. Chest CT-scans showed multiple cavitary peripheral nodules associated with lung abscesses, and in case 1 wedge-shaped densities caused by septic infarcts (Figs. 3-6). Acute onset of respiratory distress occurred on case 3 during the hospitalization and rapidly resulted in hypoxemia. Immediate chest radiograph demonstrated right pneumothorax after which an intercostal chest tube was inserted (Fig. 7). Besides pulmonary septic emboli (cases 1, 2, 3), other embolic phenomena or metastatic foci were conjunctival hemorrhage (case 1), renal infarct (case 3), cutaneous vasculitis (case 3), popliteal-femoral venous thrombosis (case 2, 4). The four patients were HIV negative; hepatitis C, HCV positive (case 2, 3, 4) and hepatitis B, HBV positive (case 3). Transthoracic echocardiography was performed in all cases while transesophageal echocardiography (TEE) was also performed in two cases (case 1, 4). All the cases have hepatosplenomegaly confirmed in abdominal ultrasound. In the acute phase, medical management consisted only in antibiotic therapy based on antibiogram. In addition to the treatment given to us by an infectious disease physician, the cardiological and toxicological treatment was equally important. For all cases, after the treatment we repeated a transthoracic echocardiography. In the cases 1 and 4 valvular vegetations disappeared (Fig. 8). In the cases 2 and 3 they persisted, so the patients underwent surgery, but not in our country. Case 3, because of acute renal failure and severe anemia, did dialysis and transfusion. Although he had multiple vegetations in the tricuspid valve and severe tricuspid regurgitation with the improvement of cardiac function, he didn’t undergo surgical intervention because of impossibility of doing this kinds of operations in our hospital. Prognosis was good for all, with 0% mortality.
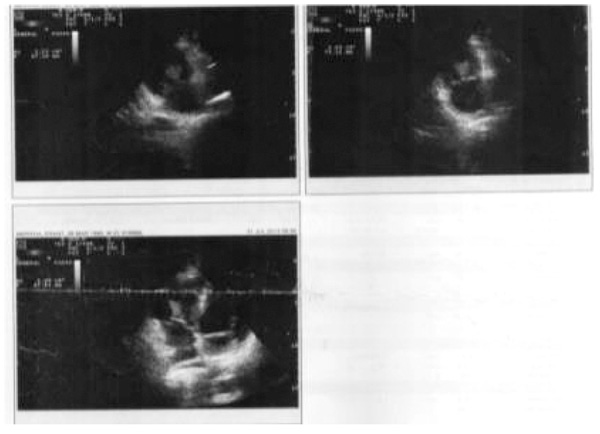 |
Fig. (1). Case 1. Large vegetation, approximately 2.7cm2. |
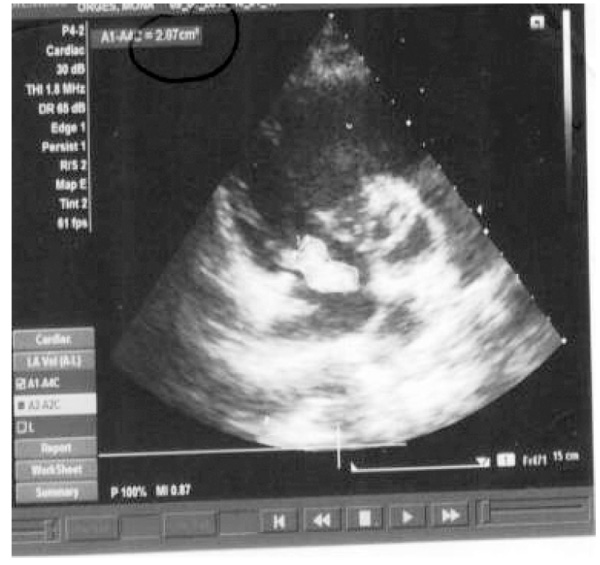 |
Fig. (2). Case 2. Large vegetation, approximately 2.87cm2. |
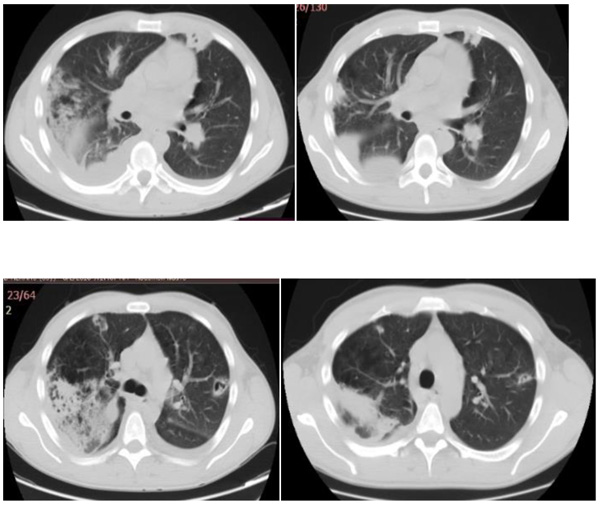 |
Fig. (3). Case 1, CT showed pulmonary wedge-shape consolidations. |
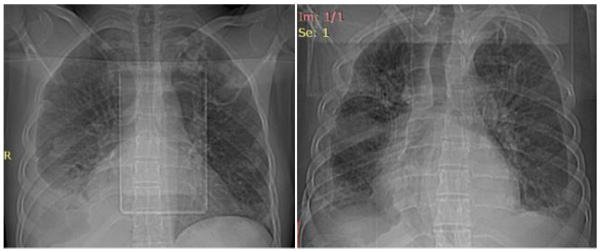 |
Fig. (5). Case 3, Radiograph showed right pleural effusion. |
 |
Fig. (8). Case 1. Disappearance of large vegetation in diameter, attached to the tricuspid valve after antibiotic treatment. |
4. DISCUSSION
Our study focused on investigating the clinical features of IE in four drug users. They were all heroin users, although the literature speaks for higher risk to cocaine users [5]. Our patients were young men from 24 to 33 years old. In keeping with the demographic characteristics of injection drug use, this disease occurs most commonly in young males in their 20s and 30s [5]. IE should be suspected in any patient with unexplained fevers, night sweats, or signs of systemic illness, particularly if any of the following risk factors are present [6]. Among injection drug users presenting with fever, 13% will have echocardiographic evidence of IE [7]. In drug users, injection of microorganisms or particulate matter from the skin itself or from within the drug material may generate transient or permanent endothelial damage to the tricuspid valve, thus providing an area for vegetations to develop. The tricuspid valve is mainly involved [8]. Endocarditis in people who use injection drugs is likely to be right-sided; therefore, septic pulmonary emboli are common, whereas manifestations of endocarditis (e.g., splinter and conjunctival hemorrhages) are less likely [9]. All our cases had tricuspid valve affection and three of them presented with pulmonary complaints and chest radiographs and CT-scans showed multiple septic pulmonary emboli. These septic emboli had all imaging features to be diagnosed as such. They were peripheric, cavity nodules with clearly identifiable feeding vessels, called feeding vessel sign [10]. In case 1 there were evident wedge-shaped densities caused by septic infarcts. They were about 15 mm, the same range as prescribed in other studies [11]. Case 3, during the hospitalization, developed pneumothorax. It was thought to be a spontaneous pneumothorax, because he had not undergone any invasive procedure before its occurrence. Pneumothorax is a possible lethal complication of septic pulmonary embolism in intravenous drug users with right-sided endocarditis and should be considered in such patients when respiratory distress occurs acutely during their hospitalization [12]. It must be due to progression of septic pulmonary infiltrates with subsequence leakage of air in the pleural cavity. Currently, between 40% and 90% of intravenous drug users with IE are HIV infected, and the HIV epidemic has caused a decrease in the incidence of this disease, probably due to changes in drug administration habits undertaken by addicts in order to avoid HIV transmission. In our case, all the patients were HIV negative and three cases (case 2, 3,4) were HCV positive. In Albania, HCV infection is more common than HIV among People Who Inject Drugs (PWID). The 2011 Bio-BSS reported HCV prevalence at 28.8% among this high-risk group. The data from laboratory surveys of HBV among PWID in 2003, 2006/07, 2009 and 2011 demonstrated prevalence rates of the surface antigen of the hepatitis B virus (HBsAg) at 10.1% (8 out of 79), 22.8% (38 out of 166), 20.2% (20 out of 99) and 23% respectively. The estimated prevalence of HIV among people who inject drugs is 0.5%, while around 28.8% are infected with HCV [13]. IE is often the result of acute S aureus infection of right-sided heart valves [14]. Blood cultures should be obtained before initiation of antibiotic therapy. Blood culture positive for S. aureus was identified in all of cases. In the cases 1, 2 and 3 we obtained more than three hemocultures, respectively 4, 6 and 7. Staphylococcal endocarditis in injection drug users is now the dominant form of the disease in many urban communities where there is a high incidence of injection drug use and homelessness. Case 4 had polymicrobial infection (Staph. Aureus, Acinetobacilis baumani,
Strept.pneumonie). This fact is seen in 15% of cases [15]. Urinalysis may show evidence of gross or microscopic hematuria, proteinuria, or pyuria. White blood cell count may be normal or elevated. All the cases had leukocytosis and case 3 had proteinuria (6.6g/l/24 h). Baseline electrocardiography should be performed in patients with infectious endocarditis so that new cardiac manifestations can be recognized early (e.g., extension of valvular disease into the conduction system, ischemia secondary to emboli to the coronary circulation) [16]. We did electrocardiography to all our patients and case 3 had changes related to severe tricuspid regurgitation. The American College of Cardiology and the American Heart Association recommend that echocardiography should be performed to identify valvular abnormalities in all patients in whom there is moderate or high suspicion of endocarditis [17]. Transthoracic echocardiography is usually the initial imaging modality. However, transesophageal echocardiography may be necessary in some patients, such as those with staphylococcus bacteremia, limited transthoracic windows because of obesity or mechanical ventilation, a prosthetic valve that renders visualization difficult secondary to shadowing, a history of endocarditis, or a structural valve abnormality. We did transthoracic echocardiography to all our patients and transesophageal echocardiography to 50% of them. Tricuspid vegetations are often large and may be in excess of 2 cm [5]. In our cases, echocardiography showed the presence of tricuspid vegetations that ranged in size from 2cm2 to 2.8cm2. The choice of definitive antibiotic therapy is based on the causative microorganism and its antibiotic susceptibility, and whether the involved valve is native or prosthetic. Our patients had native valve and right sided endocarditis. The structural and functional integrity of cardiac valves may be damaged by infection. This may lead to valvular regurgitation or flow obstruction in valves with large vegetations [6]. Surgical intervention should be considered in patients with fungal infection, infection with aggressive antibiotic-resistant bacteria or bacteria that respond poorly to antibiotics, left-sided infectious endocarditis caused by gram-negative bacteria, persistent infection with positive blood cultures after one week of antibiotic therapy, or one or more embolic events during the first two weeks of antibiotic therapy [18]. Surgical intervention is warranted for valve dehiscence, perforation, rupture or fistula, or a large perivalvular abscess. Cases 1 and 4 had total clinical and imaging improvement with a disappearance of valvular vegetations. Case 2 and 3 had clinical improvement, but without disappearance of valvular vegetations. They both underwent surgery, but because of the impossibility of realization of these kinds of operations in our country, they did it abroad. However, it is worth saying that 50% of cases had a very good prognosis. This fact is also noted in literature [19].
CONCLUSION
IE occurs in a wide range of underlying conditions where important are intravenous drug users. It is an infection that can develop dramatically over a few days. A high degree of suspicion is required and early echocardiography is recommended in patients with unexplained fever and pulmonary events. Echocardiography remains the main modality and should be used serially to facilitate early diagnosis. Coexistent medical problems such as hepatitis may be present. The successful management of a complicated case often requires the close cooperation of an infectious disease physician, a cardiologist, an addiction physician and occasionally a cardiac surgeon. This population of patients is a challenge for healthcare system because care for them is expensive and prolonged. A sharp and early attention to them avoids a huge health and financial burden.
LIMITATIONS
This is a retrospective study. The lack of a computerized system for the patient medical data and a PACS (Picture Archiving and Communication System), made the analysis difficult.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
No animals/humans were used for studies that are the basis of this review.
CONSENT FOR PUBLICATION
A written informed consent was obtained from all patients when they were enrolled.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.