All published articles of this journal are available on ScienceDirect.
Strongyloides Infection in a Man with Abdominal Pain and a History of Rheumatoid Arthritis
Abstract
Background:
Strongyloidiasis is self-limited by the complete immune system, it may be complicated and causes hyperinfection in immunocompromised patients. Objective: Here, we report a case of an immunocompromised patient with duodenal involvement of Strongyloides stercoralis.
Case Report:
A 65-year-old man presented with severe pain in central abdomen and periumbilical regions. He had no history of alcohol consumption, smoking and surgery but the history of RA (Rheumatoid Arthritis) and hypothyroidism taking immunosuppressive medications. The patient underwent endoscopy and colonoscopy which pathological analysis of the biopsies revealed remarkable findings in favor of Strongyloidiasis. After two consecutive day’s consumption of ivermectin 200µg/kg, the symptoms were completely removed. Also, the stool examination was negative for S. stercoralis larvae two weeks after end of the treatment.
Conclusions:
It seems that in immunocompromised patients with gastrointestinal symptoms assumption of parasite-like infections such as Strongyloidiasis should be considered as one of the diagnosis options. Due to the physiological and gut microbial alternations, these patients are more susceptible to infectious diseases.
1. INTRODUCTION
Strongyloidiasis which approximately infects 100 million people worldwide is a parasitosis caused by intestinal nematode Strongyloides stercoralis with the common cause of abdominal pain and diarrhea [1, 2]. Though this infection is self-limited by the complete immune system, it may be complicated and cause hyperinfection in immunocompromised patients [3, 4]. Also, it seems that the interactions of gut microbiome [5-7], pathological mechanisms, host defense pathways and effect of systemic corticosteroids [8] impacts on its manifestations. There are several desirable parasitological diagnostic methods in detection of S. stercoralis in particular among immunosuppressed patients such as serological and molecular tools [9]. As S. stercoralis colonize in the duodenum, in certain situations the diagnosis by endoscopy and biopsy may be essential for the diagnosis of this parasitosis thus reducing the risk of death of the hosts in cases of immunosuppression [10-12]. Here, we report a case of an immunocompromised patient with involvement of the duodenum by S. stercoralis.
2. CASE REPORT
A 65-year-old male with a history of RA (rheumatoid arteritis) and hypothyroidism taking immunosuppressive medications such as corticosteroid and hydroxychloroquine was admitted to the Internal Medicine Department of Razi Hospital in Rasht (a city in northern Iran). About 3 weeks ago, severe pain appeared in the central abdomen and periumbilical region which its intensity was not altered with feeding, defecation, and positional changes. A decreased appetite and constipation were added to the symptoms. Also, he complained of repeated vomiting containing the eaten food. In addition, he had about 4-5 kg weight loss. He had no history of alcohol consumption, smoking, and surgery. Clinical examinations of pulmonary were normal.
Initial blood laboratory tests revealed WBC: 8.3×103/UL (4-10 ×103/UL), MCV: 89.9 (80-100 FL), MCHC: 30.6 (31-37 gr/dL), PLT: 613×103/UL (150-450 ×103/UL), Ferritin: 610 (12-300 ng/mL), Alb: 3 (3.5-5 g/dL), K: 2.4 (3.5-5.3 mEg/L), Na: 122 (135-145 mEg/L), ESR: 21 (0-22 mm/hr), CRP: 3+ (<6 mg/L), Bill T: 1.1(0-1.1 mg/dL), Bill D: 0.3 (0-0.7 mg/dL), Cr: 16 (0.8-1.3 mg/dL), Total Pr: 5.1 (6.6-8.8 gr/dL), Ca: 6.9 (8.5-10.5 mg/dL), Fe: 64 (13.5-17.5 g/dL), TIBC: 131(240-450 mcg/dL).
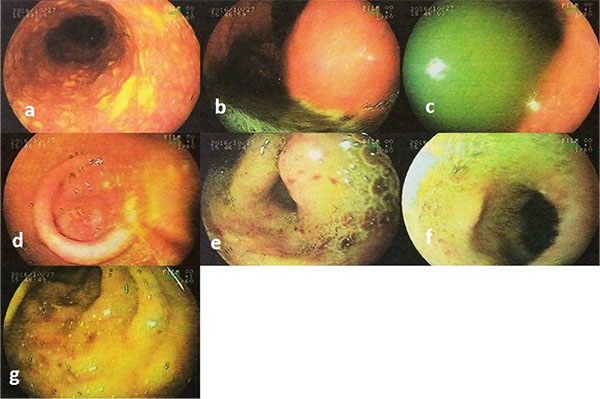
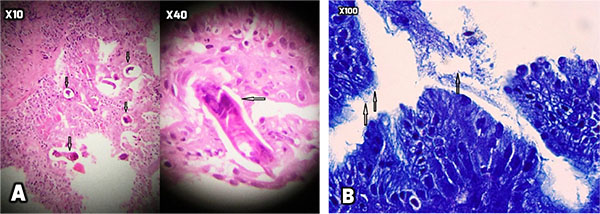
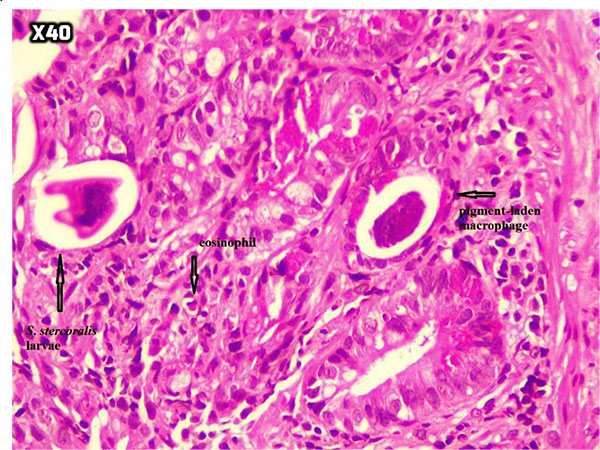
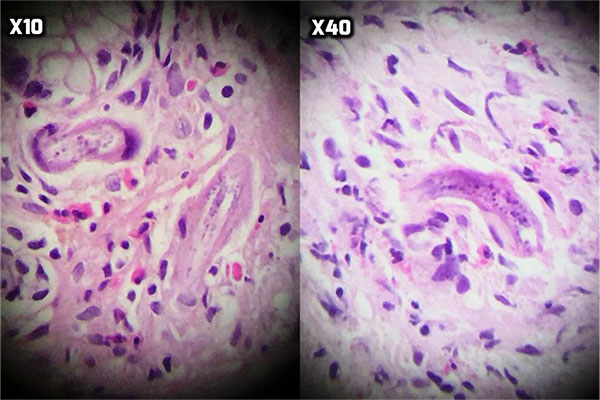
At the next step, endoscopy showed esophageal candidiasis (Fig. 1a), bile secretion in the stomach (Fig. 1b and 1c), severe erythema in the body and antrum (Fig. 1c and 1d) as well as multiple ulcers in D1 (proximal horizontal 5 cm beginning with the 3-cm duodenal bulb) and D2 (descending) (Fig. 1e, f, g). In the colonoscopy, the internal hemorrhoid grade 1, multiple diverticula in the sigmoid cecum and severe erythema with multiple ulcerations extended from descending colon to the cecum was observed (Fig. 2). During endoscopy and colonoscopy the biopsies where taken. The pathological analysis of three distinct biopsies including gastric, colonic and duodenal mucosa revealed remarkable findings. The gastric mucosa showed focal erosion, surface irregularity with mixed inflammatory cells infiltrated in lamina propria. Focal erosion, surface irregularity with mixed inflammatory cells infiltrated in lamina propria demonstrated in gastric mucosa. Furthermore, some S. stercoralis larvae were found in gastric pits with the mild colonization of H. pylori (Fig. 3). The colonic mucosa sample revealed ulceration and granulation tissue formation with focal crypt distortion, cryptitis, and focal crypt abscess formation. The lamina propria contained an increase in inflammatory cells rich in eosinophil and some pigment-laden macrophages as well as some parasite-like structures (Fig. 4). The last biopsy which was from duodenal mucosa showed the infiltration of mixed inflammatory cells in lamina propria and few S. stercoralis larva in crypts (Fig. 5).The diagnosis was strongyloidiasis according to the gastric and duodenal mucosal biopsy (D2), while the colonic mucosa biopsy just stated mucosal ulceration and eosinophil-rich in chronic active colitis, parasite-like structures, and deposition of pigment. The patient was taken ivermectin 200µg/kg for two consecutive days and the symptoms were totally removed. Also, the stool examination was negative for S. stercoralis larvae two weeks after end of the treatment.

3. DISCUSSION AND CONCLUSION
S. stercoralis Infection has a long past history [13]which mostly is linked to the suppressed status of the immune system [14, 15]. The increased worms are detectable in extraintestinal sites, especially the lung in hyperinfection syndrome [16] which involves patients with impaired cellular immunity such as those receiving long-term steroids, HTLV-1 and HIV-infected patients, and those with hematologic malignancies, transplant recipients [16]. COPD which its exacerbations might be mimicked by pulmonary strongyloidiasis is the most reason of corticosteroids receiving in patients who develop hyperinfection syndrome [16-18] The occurrence of hyperinfection syndrome may be as early as 20 days after the onset of corticosteroid therapy [19] and as late as several years [20].
A combination of gastrointestinal, pulmonary and constitutional symptoms are the common manifestations. Also, severe cramping, abdominal pain, watery diarrhea, weight loss, nausea, vomiting, occasionally gastrointestinal bleeding and small bowel obstruction are considered as intestinal manifestations [3]. Asthma-like symptoms such as cough and wheezing, and others such as pneumonia, pulmonary hemorrhage, pleural effusion, and acute respiratory failure are founded as its pulmonary manifestations [3, 21]. Hyperinfection may be complicated by bacterial infections and bacteremia caused by gut flora [7].
Although the diagnosis of strongyloidiasis is definitely based on larvae detection in stool or sputum [16, 22], it is not as adequate as other methods such as blood agar plate culture [16, 23], duodenal fluid sampling and serologic testing [16, 24],. However, it should be considered that depending on the serological test employed the sensitivity and specificity may be low and culminate in cross reactivity. Along with some effective drugs such as albendazole, mebendazole, thiabendazole, and ivermectin for S. stercoralis infection according to the more studies in the treatment of strongyloidiasis, ivermectin is the drug of choice [25, 26]. Monitoring the response to treatment could be very difficult with detection of S. stercoralis larvae in the stool specimen because of the inconsistent shedding of the larvae.
In our case which was suspected of hyperinfection syndrome, in contrast to other reported cases, there were no pulmonary manifestations. However, the gastrointestinal manifestations according to the gastric and colonic biopsies lead us to diagnose this infection. As regard as, biopsy is not preferable to an invasive method there was no way to distinguish of this by stool examination or bacterial culture. As it is mentioned that it may be unreasonable to screen immunocompromised patients from endemic areas by serology before initiating steroid therapy to prevent the development of Strongyloides hyperinfection, screening is not recommended for patients before starting short courses of corticosteroids for bronchial asthma or COPD, but it may be considered in severe cases of bronchial asthma or COPD requiring recurrent and frequent steroids courses [27].
According to our case, it is recommended to notice that the physicians should consider the suspicion of Strongyloides in immunosuppressive patients. This patient who had RA along with hypothyroidism the reduction of metabolism and complicated alterations of gut microflora may explain the hyper infection. As recently it is stated that microbiome has an important impact on the cellular interactions in which its weakness may explain the cause of S. stercolris. Also, it seems that in accordance with the other studies ivermectin is still an appropriate choice of treatment. The patient was cured without any relapse according to the negative stool examinations in a two-week follow-up.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
No animals/humans were used for studies that are the basis of this research.
CONSENT FOR PUBLICATION
A written informed consent was obtained from all the patients when they were enrolled.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
We would like to thank all the members of Gastrointestinal & Liver Diseases Research Center (GLDRC). We also thank the patient’s family for providing consent for this case report.


