All published articles of this journal are available on ScienceDirect.
Original Versus Generic Direct Acting Antivirals in Treatment of Chronic Hepatitis C Patients: Real Life Data From Latvia
Abstract
Background:
Despite effective treatment available from 2016 in Latvia, there are restrictions - only patients with fibrosis (F, Metavir) stage 3 to 4 have access to reimbursed medicines. Some patients obtain generic drugs from India.
Objective:
The aim of this study was to evaluate the efficacy of original and generic direct acting antiviral medications in Latvian patients.
Materials and Methods:
This is a retrospective study of 179 chronic virologic hepatitis C patients. Data were obtained from patients’ medical records. Mean age 49.2 years (SD 10.2, range 24-71), 88 female patients (49%), 91 male patients (51%). Genotype 1b was detected in 157 patients (87.7%). Patients were divided into two groups - patients who received original direct acting antivirals ombitasvir, paritaprevir, ritonavir, dasabuvir + ribavirin (n=144, F3-4, Child-Pugh A) and those who received generic medicines from India (n=35, F0-2) sofosbuvir, ledipasvir (n=7) or sofosbuvir, daclatasvir + ribavirin (n=28). Undetectable viral load 12 weeks after cessation of therapy (sustained virologic response 12 or SVR12) was measured in all patients. Therapy course completed 142 patients from original medicines group and all patients from generics group.
Results:
In the original medicines group - sustained viralogic response was achieved in 142 patients who completed treatment course (100%), while in generic medicines group in 32 patients (91.4%).
Conclusion:
Study results show high efficacy of both regimens using original and generic medicines - sustained virologic response was achieved in more than 90% of patients, with slight superiority in original medicines group.
1. INTRODUCTION
Chronic viral hepatitis C is a global healthcare problem. In the early stage the disease progresses asymptomatically, although some patients may complain about fatigue and other unspecific symptoms. Since there are no screening rules, in most cases the disease is diagnosed at a very late stage. More than seventy one million people suffer from viraemic hepatitis C virus (HCV) infections worldwide [1] and 350,000-500,000 patients die from liver diseases associated with HCV infection each year [2, 3]. The previous interferon based therapy was less effective and caused many side effects. Recently - many new and effective medicines were introduced. There are many advantages, including oral use, shorter treatment courses and fewer side effects, and most importantly - more than 90% of patients are reaching sustained virologic response [4].
According to the epidemiological study carried out in 2008, there were more than 39 thousand patients with chronic hepatitis C in Latvia [5]. Similarly, to majority of countries worldwide Injection Drug Use (IDU) and a historically unsafe blood supply were major risk factors for acquiring HCV infection [6]. Many chronic hepatitis C patients have lower quality of life comparing to general population.
According to the data provided by the Centre for Disease Prevention and Control in Latvia, there were 1907 patients with newly diagnosed chronic hepatitis C in 2016 comparing to 1789 in 2015 [7]. The growing trend could be due to many reasons, for example, better HCV infection awareness in the population and especially among persons representing risk groups, better diagnostics and the assumption that HCV infection continues to spread at the same pace as previous 5-10 years.
The number of newly diagnosed cases of acute hepatitis C does not decrease as well. Acute viral hepatitis C is asymptomatic in most cases and rarely is associated with life threatening health condition. Spontaneous cure within 6 months in the absence of any treatment is observed in approximately 15-45% of cases. In the remaining cases, chronic hepatitis C may develop. The risk of developing liver cirrhosis in 20 years’ period is 15-30% [8]. It is very likely that the burden of advanced liver disease outcomes will increase in the future as the prevalent population ages and their disease progresses [9, 10]. But it is possible to reduce disease burden substantially by improving diagnosis and treatment. Especially important is the use of medicines with improved treatment efficacy [11]. World Health Organization (WHO) has set clear targets for managing HCV infection in the new era of Direct Acting Antiviral (DAA) medicines. Being part of European Union (EU), Latvia is well positioned to achieve those targets namely a 65% reduction in liver-related deaths, a 90% reduction of new viral hepatitis infections, and 90% of patients with viral hepatitis infections to be diagnosed by 2030 [12]. Patient access to DAA medicines will be the key factor in the coming years and there are recommendations for countries to follow the 2016 hepatitis C virus treatment guidelines issued by the European Association for the Study of Liver [13].
Direct acting antiviral medicines ombitasvir, paritaprevir/ritonavir, dasabuvir are 100% reimbursed in Latvia as of January 1st 2016. Elbasvir, grazoprevir was included into reimbursement list from January 1st 2017, but all patients do not have access to this highly effective treatment. There are strict reimbursement criteria such as - only patients with HCV genotype 1 or 4 infection and fibrosis (F, Metavir) of stage F3 and F4 were eligible at the beginning. Starting from February 1st 2018 patients infected with HCV of all genotypes are eligible for reimbursed treatment. In addition - patients before or after solid organ transplantation, those with clinically significant extrahepatic manifestations, for example chronic kidney failure requiring dialysis, and women before carrying out in vitro fertilization also can receive reimbursement for treatment with new DAA medicines [14]. As of February 1st 2018, there is only one combined DAA medicine reimbursed - sofosbuvir/velpatasvir.
Remaining group of chronic hepatitis C patients which have no access to State reimbursed treatment with newer medicines, for example, those with fibrosis stage F0, F1 and F2 either stay without treatment or choose to obtain generic medicines directly from India.
Most common medication side effects include nausea, headache, insomnia, fatigue, itching, other skin infections, increased levels of bilirubin and decreased hemoglobin levels. The side effects are encountered more often if ribavirin is added to the scheme. The increase of alanine aminotransferase (ALT) level in clinical studies was observed in 1% of the patients. The ALT level stabilized in two to eight weeks. Increase of ALT is most commonly encountered between women that have used the oral contraception [15].
2. MATERIALS AND METHODS
A retrospective study was performed in Riga East University Hospital, Latvian Center of Infectious Diseases. Data was obtained from patients’ medical records in the period from November 2016 to March 2017. Adult patients with diagnosis “Chronic viral hepatitis C” who received DAA’s treatment where included. Many patients received the medications reimbursed by the state (ombitasvir, paritaprevir/ ritonavir, dasabuvir + ribavirin), while other patients - non-reimbursed generic medications obtained from India (sofosbuvir, daclatasvir or sofosbuvir, ledipasvir + ribavirin).
Information about the patients was summarized in the table (MS Excel 2013). The following data were collected:
- Gender;
- Age;
- HCV genotype;
- Stage of fibrosis before treatment (biopsy or fibroscan); liver biopsy was done before treatment and was base on Metavir scoring system; fibroscan results where interpreted according to FDA approved values: <7.0 kPa (F0-1), >7.0 kPa (F2), >9.5 kPa (F3), >12.0 kPa (F4);
- Treatment schedule;
- Duration of the therapy;
- HCV-RNA results at the end of therapy;
- HCV-RNA results 12 weeks after completing treatment (SVR12);
- Previous treatment;
- Child-Pugh score of the patients with cirrhosis.
Patients were divided into 2 groups - patients who received original DAA’s ombitasvir, paritaprevir/ritonavir, dasabuvir + ribavirin (n=144, F3-4, Child-Pugh A) and patients who received generic DAA’s from India (n=35, F0-2) sofosbuvir, ledipasvir (n=7) or sofosbuvir, daclatasvir + ribavirin (n=28). SVR 12 was measured in all patients.
The statistical processing of data was carried out in program SPSS 22.0, using Fisher’s exact test. Results were considered to be statistically significant, if probability was sufficiently low (p<0.05).
Patient characteristics (Table 1) - there were 179 patients included in the study, mean age 49.2 years (SD 10.2, range 24-71), 88 female patients (49%) and 91 male patients (51%). Patients with co-infections of hepatitis B virus (HBV) and human immunodeficiency virus (HIV) were excluded from the study.
| Patients’ Characteristics | n (%) | |
|---|---|---|
| Genotype | 1b | 157 (87.7) |
| 1 (subtype not known) | 8 (4.5) | |
| 2 | 2 (1.2) | |
| 3 | 9 (5.0) | |
| 1b/4 | 3 (1.7) | |
| Fibrosis Stage (Metavir) | 0 | 6 (3.4) |
| 1 | 20 (11.2) | |
| 2 | 5 (2.8) | |
| 3 | 74 (41.3) | |
| 4 | 74 (41.3) | |
Most of patients (n=102 or 57% from total) received combination therapy with 4 medicines (ombitasvir, paritaprevir/ritonavir, dasabuvir) while 42 patients - 5 antivirals (ombitasvir, paritaprevir/ritonavir, dasabuvir plus ribavirin). Combination of two drugs sofosbuvir/daclatasvir or sofosbuvir/ledispavir were given to 27 (15.1%) and 7 (3.9%) correspondingly while only one patient received combination of three medicines - sofosbuvir, daclatasvir and ribavirin.
Most patients (173 or 96.6% from total) received antiviral therapy for 12 weeks while longer therapy of 24 weeks was given to 4 patients. Therapy was discontinued in 2 patients.
The aim of this study was to evaluate efficacy of original versus generic direct acting antiviral medications. Efficacy was measured using SVR12. HCV-RNA was measured using real time PCR, Abbott real time HCV, Abbott Molecular Inc., quantitative limits from 12 IU/ml to 100 million IU/ml.
Study was designed according to Declaration of Helsinki, approved by Ethics board of University of Latvia.
3. RESULTS
Excellent treatment results were achieved in patients who completed therapy (Chart 1). Two patients (1.4%) in the original DAA group discontinued treatment due to decompensation of liver cirrhosis or side effects. One patient receiving ombitasvir, paritaprevir/ritonavir, dasabuvir stopped treatment earlier because of jaundice, upper abdominal pain on the right side, fever and swelling of face and legs a few days after starting treatment. Laboratory tests showed increased levels of ALT and bilirubin. This patient had 1b genotype, liver cirrhosis, Child - Pugh A class. Second patient on ombitasvir, paritaprevir/ritonavir, dasabuvir plus ribavirin treatment stopped treatment earlier because of effects - itchy rash that spread throughout the whole body, as well as skin hyperemia that was accompanied by fever.
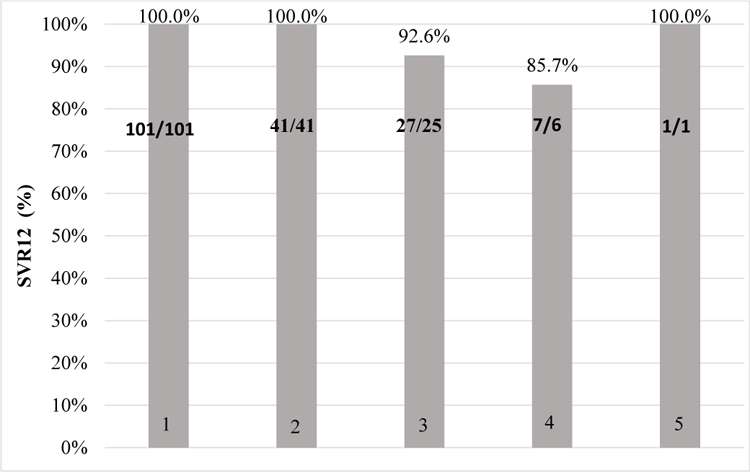
- Ombitasvir, paritaprevir/ritonavir, dasabuvir
- Ombitasvir, paritaprevir/ritonavir, dasabuvir + ribavirin
- Sofosbuvir, daclatasvir
- Sofosbuvir, ledipasvir
- Sofosbuvir, daclatasvir + ribavirin
Ombitasvir, paritaprevir/ritonavir, dasabuvir full treatment course completed 101 patients. HCV RNA 12 weeks after cessation of treatment was not detected in 100% (n=101).
Ombitasvir, paritaprevir/ritonavir, dasabuvir + ribavirin full treatment course completed 41 patients. SVR 12 was achieved in 100% (n=41).
Daclatasvir and sofosbuvir combination treatment received 27 patients. HCV RNA 12 weeks after cessation of treatment was not detected in 92.6% (n=25). Therapy was not effective in 3.7% (n=1) and HCV infection relapsed in 3.7% (n=1) of cases.
Ledipasvir and sofosbuvir combination treatment receveid 7 patients. HCV RNA 12 weeks after cessation of treatment was not detected in 85.7% (n=6), relapse observed in 14.3% (n=1).
Daclatasvir, sofosbuvir and ribavirin treatment was used in 1 patient and SVR12 was achieved in this case.
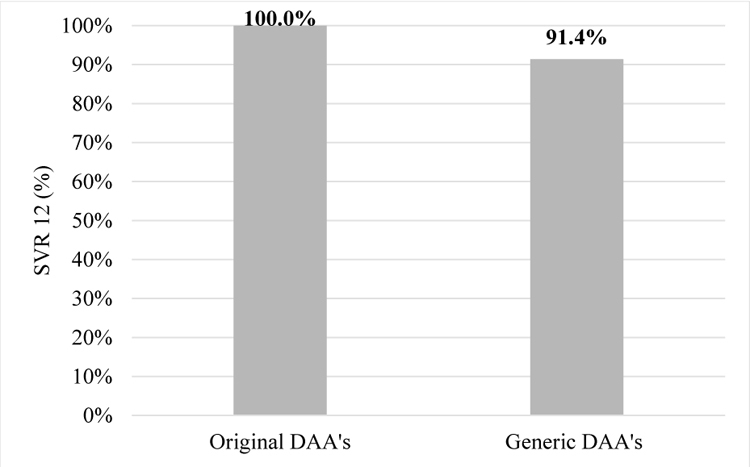
In the original DAA’s group all 142 patients achieved SVR12, but in generic DAA’s group SVR12 was achieved in 32 patients (91.4%, p=0.007, Fischer’s test).
In the generic DAA’s group SVR12 was not achieved in 3 patients (8.6%) - one patient was HCV-RNA positive right after treatment and two had relapses of HCV infection.
Comparison of treatment results in patients receiving original and generic DAA’s and who completed therapy is shown in Chart (2).The second chart shows comparison between and medicines.
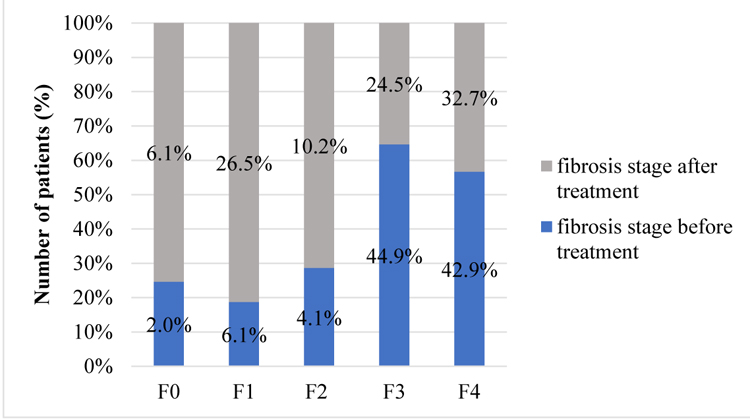
All included patients underwent liver biopsy for fibrosis staging before treatment. 49 patients had Fibroscan examination 3 to 6 months after DAA’s treatment. Before the start of therapy there were several patients with cirrhosis and fibrosis in stage 3. After the completion of treatment liver fibrosis stages 1 and 2 were detected more frequently. Fibrosis stage 1 was detected only in 6.1% of patients (n=3) before treatment and already in 26.5% (n=13) after, while fibrosis stage 2 in 4.1% (n=2) and 10.2% (n=5) respectively.
This result indicates significance of reaching the SVR. After eradication of HCV - regeneration of liver tissue is possible. There are stages of liver fibrosis before and after treatment in patients who had Fibroscan examination after completion of treatment (Chart 3).
4. DISCUSSION
The results of the research suggest that although almost all patients receiving DAA therapy achieve SVR12, there are some additional factors that influence treatment result.
Despite medications being effective, the side effects can occur. Because of the chance of side effects all patients should be carefully monitored during the period of treatment.
In the patients group receiving original medicines, the efficacy was 100% for those who completed the full treatment course. This result is similar to the data from clinical studies, where the efficacy of ombitasvir, paritaprevir/ritonavir regimen equals 100% in genotype 1b infections. It should be noted that in our study, these patients were all with liver fibrosis stages F3 or F4, because only in these cases patients receive reimbursed medicines according to the current reimbursement criteria. It is evident that even for this patient group with advanced disease - treatment efficacy is outstanding. Two patients discontinued treatment earlier - one due to decompensation of cirrhosis and the other because of pruritus. First patient’s decompensation indicators improved after discontinuation of treatment.
The treatment efficacy in the patients’ group receiving generic medicines was 91.4%. In 3 patients (8.6%) treatment was ineffective. The generic medicines were purchased out-of-pocket and delivered from India either by patients themselves or by other person. Some patients ordered medicines using the internet or postal services therefore quality of these medications is unknown.
Generic medicines are chemically equivalent to the original ones and contain the same active substance, however there might be some differences in color and size of the pills [16].
The difference in treatment efficacy between the original and generic medicines could be explained by pharmaceutical supply chain risks. While original medicines are supplied through well controlled supply chain, generic ones purchased abroad lack stringent control processes while reaching patients. Among many risks disrupting the supply chain of novel HCV medicines are pharmaceutical company internal ones, for example, human mistakes, mismanagement of processes leading to deviations in quantity and quality as well as external risks related to wholesale and retail sector. Especially dangerous activity is purchasing of medicines via Internet. Risks include counterfeit drugs as well as problems in the delivery of medicine to the right place and at the right time.
Although the efficacy of original medications reimbursed by the state and generic drugs slightly differs in this small retrospective study many patients may still be highly motivated to obtain DAA’s abroad. Risks of buying lower quality or even harmful medication seems to be perceived as lower comparing to the risk of progressing chronic disease if a case no treatment is administered.
Patients treated with State reimbursed DAAs are being closely monitored to comply with treatment guidelines and rules while patients which purchase generic products are often less controlled by a treating physician. At the same time motivation of the latter patient group could be higher taking in account personal money invested.
National reimbursement policy of DAA’s medications may need to change in Latvia and novel medicines should become available to all patients with documented chronic HCV infection not just for those with liver fibrosis stages F3 or F4.
The main study limitation is that study groups are not 100% equal, in original DAA’s group there were more patients with F3 or F4 with genotypes 1 and 4, but in generic group - F0-2, different genotypes. This is due to the reimbursement restrictions in Latvia. But still - the efficacy trend is visible in both groups.
Also, we have to take into account that normally generic drugs are registered and tested before that, but in our case patients seek for solution - for treatment of hepatitis C when it is not reimbursed by the state, and they are getting generic drugs not registered in EU.
CONCLUSION
The study results indicate not only clearance of virus after treatment but also regression of fibrosis which is very important with regard to decreasing risk of liver cirrhosis, decompensation and hepatocellular carcinoma. Further clinical trials are necessary to strengthen this evidence.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was approved by Ethics Board of University of Latvia.
HUMAN AND ANIMAL RIGHTS
This study was designed according to The Declaration of Helsinki.
CONSENT FOR PUBLICATION
Not applicable.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


