All published articles of this journal are available on ScienceDirect.
Clarithromycin versus Amoxicillin alone or with Clavulanate in Acute Maxillary Sinusitis: A Meta-analysis of Clinical Trials
Abstract
Objective:
A meta-analysis was performed to explore the relative effects of clarithromycin and amoxicillin (with or without clavulanate potassium) in the treatment of acute maxillary sinusitis.
Methods:
Six studies were identified in the peer-reviewed literature. All were randomized single-blind (investigator-blind) or open-label trials in outpatients diagnosed with acute maxillary sinusitis. A total of 1580 patients were enrolled, of whom 1194 were clinically evaluable.
The total daily dose of clarithromycin was 1000 mg; the total daily dose of amoxicillin (with or without clavulanate potassium) was either 1500 or 2000 mg. The duration of study drug treatment varied from 8 to 14 days. Endpoints comprised clinical and radiological success within 48 h of the end of study drug treatment plus bacteriologic cure and eradication.
Success and cure rate differences were analyzed using fixed- and random-effect models. The absence of between-study heterogeneity was tested using Cochran’s Q-test.
Results:
Clinical success rates varied between 85.8% and 97.9% for clarithromycin and between 84.2% and 96.8% for amoxicillin. The combined rate difference in clinical success rates between clarithromycin and amoxicillin was +1.9% (P=0.14). Radiological success rates (four studies) varied from 78.2% to 94.0% for clarithromycin and 79.7% to 95.0% for amoxicillin, with a combined rate difference of zero (P=1.00). Bacteriologic cure rates (four studies) were 87.1–94.6% for clarithromycin, compared with 89.8–98.1% for amoxicillin, with a combined difference in cure rates of –3.2% (P=0.16). Overall bacterial eradication rates were comparable between the two treatments (clarithromycin, 89.3%; amoxicillin, 92.1%).
Conclusion:
These data, with their limitations properly acknowledged, identify clarithromycin as a valid and viable alternative to amoxicillin for the treatment of acute maxillary sinusitis in adults.
1. INTRODUCTION
Maxillary sinusitis is the inflammation of the paranasal sinuses caused by an infectious agent, such as a virus, bacte- rium, or fungus, or as the result of an allergic reaction. Cases may present with some degree of ethmoid involvement.
This apparently relatively insignificant condition can have a significant socio-economic impact, much of it through indirect costs arising from work absences [1]. Estimates from a prevalence-based cost-of-illness study suggest that >$3.5 billion is spent annually on treatments, including possibly $600 million on antibiotics [2]. Untreated sinusitis may evolve into complications such as meningitis, brain abscess, orbital invol- vement, and cavernous-sinus or cortical-vein thrombosis.
We have previously reported the results of a concise meta-analysis that explored the effects of clarithromycin in the management of pharyngitis caused by Group A beta-hemolytic Streptococcus strains (GAS) [3]. Our overall conclusion from that investigation was that although clarithromycin, a second-generation macrolide that has an expanded spectrum of activity and tolerability relative to erythromycin [4], is currently not specified as first-line therapy for upper respiratory tract infec- tions in many guidelines, it may nevertheless be an effective and largely well-tolerated treatment for GAS pharyngitis patients, and can be considered as an alternative treatment option [5].
In the course of that exercise, we identified a group of studies comparing clarithromycin and amoxicillin in the treat- ment of Acute Maxillary Sinusitis (AMS). We now summarize the findings of our appraisal of those studies.
2. MATERIALS AND METHODS
Relevant studies were identified by reference to the 2014 Cochrane Collaboration review by Ahovuo-Saloranta and colleagues [6] and corroborated and supplemented by PubMed searches framed around the terms ‘clarithromycin,’ ‘amoxici- llin,’ ‘sinusitis,’ and ‘clinical trial.’ No a priori language limita- tion was imposed. Study quality was quantified using the Jadad score scale (Appendix 1), in which possible scores range from zero (worst) to five (best) [7].
Endpoints were clinical and radiological success as evaluated within 48 h after the end of study drug treatment and bacteriologic cure and eradication rates. Clinical success was defined as either cure (signs and symptoms resolved) or improvement (signs and symptoms improved but not resolved). Radiological success was defined as either resolution or improvement. Pathogens were considered eradicated if none could be cultured after the end of study drug treatment or if there was no clinical indication for culturing. A bacteriologic cure was defined as the eradication of all pathogens.
To obtain combined estimates with Confidence Intervals (CIs) for the differences in success and cure rates between clarithromycin and amoxicillin, either a random-effects model based on the DerSimonian−Laird method or a fixed-effect model based on the inverse-variance method was performed, with the rate difference as the effect measure [8]. The absence of between-study heterogeneity was tested using Cochran’s Q-test.
3. RESULTS
3.1. Studies
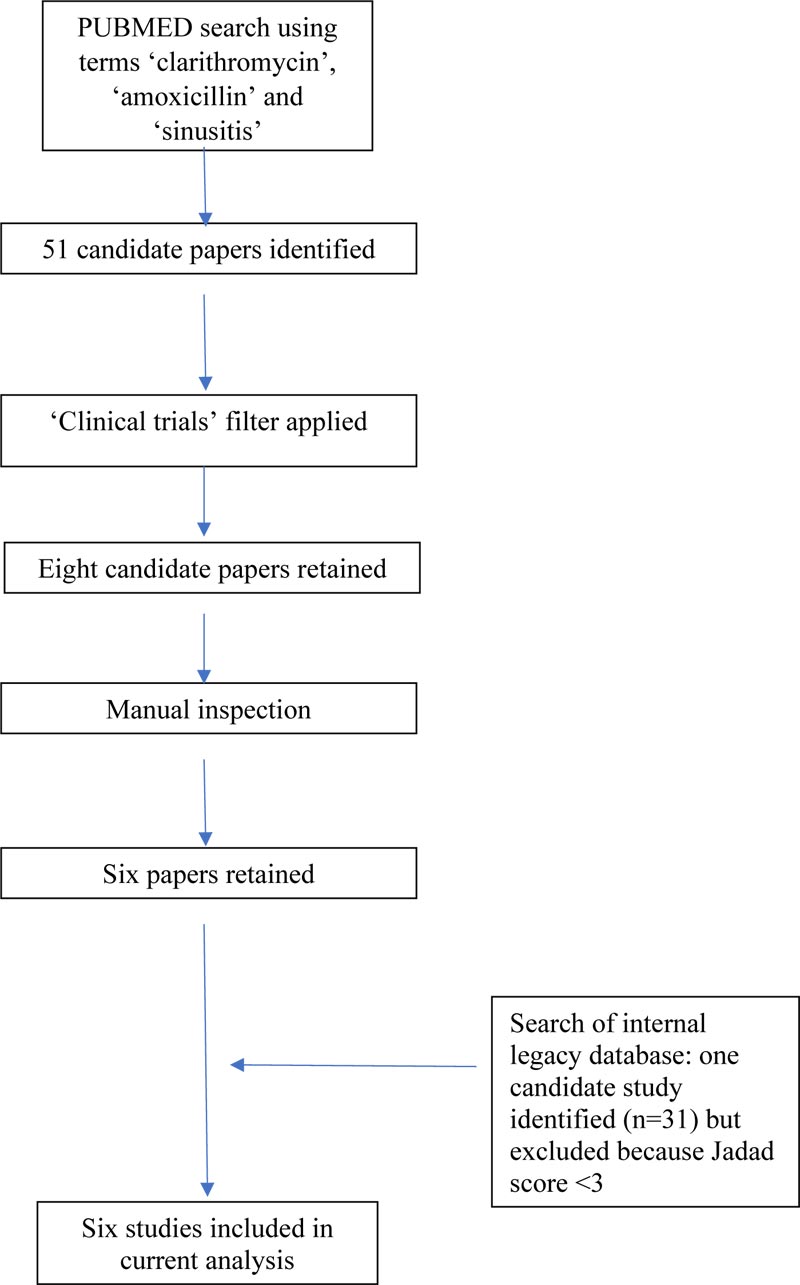
Six studies were identified in the peer-reviewed literature [9-14]. The course of identification of papers is illustrated in Fig. (1). All studies were randomized single-blind (investi- gator-blind) or open-label trials in outpatients diagnosed with AMS.
Summary particulars of the final selection of papers are presented in Table 1. In aggregate, these randomized 1580 patients. After the exclusion of 440 patients, primarily for pre-treatment failure to identify a target pathogen, 1140 were available for the evaluation of clinical response and 818 for evaluation of bacteriological response.
In all studies, the total daily dose of clarithromycin was 1000 mg, given as 500 mg b.i.d. (five studies) or 1000 mg o.d. (extended-release formulation; one study). The total daily dose of amoxicillin was either 1500 mg given as 500 mg t.i.d. (three studies) or 2000 mg, given as 1000 mg b.i.d. (two studies). The duration of study drug treatment was up to 14 days (four studies), up to 11 days (one study), or up to 8 days (one study). In three studies, the amoxicillin formulation was a fixed combination with clavulanate potassium, a beta-lactamase inhibitor that prevents certain bacteria from becoming resistant to amoxicillin.
Details of the individual studies are provided below.
3.1.1. Calhoun & Hokanson
The diagnosis of AMS had to be confirmed by a sinus roentgenogram. Results of pre-treatment susceptibility testing were not reported. One hundred and forty-two patients (58 males, 84 females) aged 14–77 years were enrolled, of whom 70 received clarithromycin and 72 amoxicillin. Twenty-six patients were excluded from the analysis, 15 in the clarithromycin group and 11 in the amoxicillin group. In the clarithromycin group, the primary reason for exclusion was non-compliance (n=6), while in the amoxicillin group, the primary reason for exclusion was premature discontinuation of therapy (n=5). Other reasons for exclusion included misdiagnosis, no pre-treatment roentgenographic confirmation of sinusitis, receiving fewer than 3 days of study drug treatment, no follow-up sinus roentgenogram, or no follow-up examination [12].
3.1.2. Dubois et al.
The diagnosis of AMS had to be confirmed by a positive maxillary sinus radiograph and positive culture of sinus fluid. Results of pre-treatment susceptibility testing were not reported. In total, 497 patients aged ≥12 years were randomized, 246 to treatment with clarithromycin and 251 to treatment with the fixed combination amoxicillin/clavulanate. A total of 103 patients treated with clarithromycin and 117 treated with amoxicillin were excluded from analysis due to the absence of pathogen in the pre-treatment culture. A further 17 patients (11 with clarithromycin, six with amoxicillin) were excluded for unspecified reasons. In the clarithromycin group, the infection was mild in 15 evaluated patients (11.4%), moderate in 113 (85.6%), and severe in four (3.0%). In the amoxicillin/clavulanate group, the corresponding numbers were 16 (12.5%), 103 (80.5%), and nine (7.0%), respectively [11].
3.1.3. Géhanno et al.
Patients suspected of suffering from AMS had to undergo both a pre-treatment roentgenogram and a sinus culture. Susceptibility testing established that 25% of the Haemophilus influenzae isolates were producers of beta-lactamases but that all were sensitive to clarithromycin or the combination amoxicillin/clavulanate. Decreased sensitivity to penicillin was observed in 7/57 (12.3%) Streptococcus pneumoniae isolates, and 15/57 (26.3%) were deemed resistant to clarithromycin. In total, 284 patients (148 males, 136 females) were randomized, 145 to treatment with clarithromycin and 139 to treatment with the combination amoxicillin/clavulanate. Sinus infection was bilateral in 28 of the clarithromycin patients and in 20 of those who received amoxicillin. Eleven patients treated with clarithromycin and 10 treated with amoxicillin were excluded from the analysis. The clarithromycin patients were excluded either because the radiologic investigation was not done (n=4) or because it did not reveal any abnormality (n=7). Corres- ponding numbers for the amoxicillin group were three and four, respectively. Sinus infection was bilateral in 41 (30.6%) of the evaluated clarithromycin patients and in 36 (27.3%) of the evaluable patients who received amoxicillin [13].
| Reference | Clarithromycin Regimen | Comparator Regimen | Study Design | Number of Patients Randomized/Clinically Evaluated/Bacteriologically Evaluated | Sex Distribution/Age | Endpoints | Jadad Scoreb |
|---|---|---|---|---|---|---|---|
| Calhoun & Hokanson [12] | 500 mg b.i.d. for 7−14 days | Amoxicillin 500 mg t.i.d. | Randomized, multicenter, single-blind | 142/116/not applicable | Men: 58; women: 86/age range 14–77 years: mean ≈37 years | Clinical and radiological success | 3 |
| Dubois et al. [11] | 500 mg b.i.d. for up to 14 days | Amoxicillin/clavulanate 500 mg t.i.d. | Randomized, multicenter, single-blind | 497/260/294 | Clinical and radiological success; bacteriologic cure; bacteriological eradication | 3 | |
| Géhanno et al. [13] | 500 mg b.i.d. for 8 days | Amoxicillin/clavulanate 500 mg t.i.d. | Randomized, open-label | 284/263/158 | Men: 139; women: 145/mean age ≈40 years | Clinical success | 3 |
| Karma et al. [10] | 500 mg b.i.d. for 9−11 days | Amoxicillin 500 mg t.i.d. | Randomized, multicenter, single-blind | 100/68/61 | Men: 99; women: 11/age range 18–69 years: mean ≈30 years | Clinical and radiological success; bacteriologic cure; bacteriological eradication | 3 |
| Marchi [9] | 500 mg b.i.d. for up to 14 days | Amoxicillin 1000 mg b.i.d. | Randomized, multicenter, open-label | 120/114/87 | Men: 74; women: 46/age range 19–76 years: mean ≈48 years | Clinical success; bacteriologic cure | 3 |
| Riffer et al. [14] | 1000 mg o.d.a for 14 days | Amoxicillin/clavulanate 1000 mg b.i.d. | Randomized, multicenter, single-blind | 437/373/218 | Men: 194; women: 243/age range 13–79 years: mean ≈37 years | Clinical and radiological success; bacteriologic cure; bacteriological eradication | 3 |
3.1.4. Karma et al.
The diagnosis of AMS had to be confirmed by the presence of sinus fluid during antral puncture, and the pathogens cultured from the fluid had to be susceptible to both study antibiotics. Detailed results of pre-treatment susceptibility testing were not reported, but pathogens cultured from sinus fluid were required to be susceptible to both study antibiotics. One hundred patients ranging in age from 17 to 69 years (89 males, 11 females) were enrolled, of whom 50 were randomized to treatment with clarithromycin and 50 to treatment with amoxicillin. Sinus infection was bilateral in 28 (56.0%) of the clarithromycin patients and 20 (40.0%) of those who received amoxicillin. For 34 patients in the clarithromycin group and 38 in the amoxicillin group, causative pathogens could be isolated from the pre-treatment sinus fluid culture. One patient in the clarithromycin group was excluded from the analysis because of concomitant pneumonia. In the amoxicillin group, two patients were excluded because they did not have susceptibility results reported. A third patient discontinued treatment due to an adverse event (diarrhea) [10].
3.1.5. Marchi
In total, 120 patients (74 males, 46 females) ranging in age from 19 to 76 years were randomized, 61 to treatment with clarithromycin and 59 to treatment with amoxicillin. The clinical condition of the patients was rated good for all clarithromycin patients and for all but one of the amoxicillin patients, whose condition was rated fair. Results of pre-treatment susceptibility testing were not reported, but the study protocol stipulated that all pathogens were required to be susceptible to both study antibiotics. In the clarithromycin group, 57 of 61 patients were evaluable clinically, and 47 were evaluable bacteriologically. Of the four patients who were not clinically evaluable, one was diagnosed after the termination of study treatment as having chronic maxillary sinusitis, while for three patients, post-treatment signs and symptoms were not recorded. In the amoxicillin group, 57/59 patients were evaluable clinically, and 40 were evaluable bacteriologically. Post-treatment signs and symptoms were not recorded for two patients, who were therefore excluded from the analysis.
3.1.6. Riffer et al.
The diagnosis of acute, uncomplicated bacterial sinusitis was based on opacification or an air/fluid level in a sinus radiograph or computerized tomography scan of the maxillary sinus(es), purulent nasal discharge, and at least two relevant signs and symptoms lasting between 8 and 28 days before screening. A total of 437 patients in the age range 13–79 years (194 males, 243 females) were enrolled, of whom 221 were randomized to treatment with clarithromycin and 216 to treatment with the combination amoxicillin/clavulanate. Two analysis sets were defined: a clinically evaluable set and a clinically and bacteriologically evaluable set. In total, 33 clarithromycin and 31 amoxicillin patients were excluded from the clinically evaluable population. Reasons for exclusion were: selection criterion not met (n=21), no radiographic confirmation of diagnosis (n=6), non-compliance (n=5), missing endpoint data (n=18), confounding medication (n=10) and other (n=4). Excluded from the clinically and bacterio- logically evaluable set were 166 clarithromycin and 162 amoxicillin patients. Here, the major reasons were no target pathogen isolated (n=205) or aspiration/endoscopy data missing (n=88). Detailed results of susceptibility testing were not reported, but it was recorded that five of 22 isolates of S. pneumoniae isolated from patients randomized to clarithro- mycin and who were clinically and bacteriologically evaluable were classified as resistant on the basis of in vitro tests, but the pathogen was deemed to have been eradicated at the completion of treatment [14].
3.2. Isolated Pathogens
Four of the six studies reported pathogens that were isolated pre-treatment (Table 2). The most frequently reported pathogens were S. pneumoniae (31.8%), Haemophilus (28.6%), and Staphylococcus aureus (20.8%).
3.3. Clinical Success
Clinical success rates are summarized in Table 3. Success rates were high and varied between 85.8% and 97.9% for clarithromycin and between 84.2% and 96.8% for amoxicillin. The heterogeneity of success-rate differences among studies was non-substantial (Cochran’s Q=3.6, P=0.60), justifying a fixed-effect meta-analysis. The combined difference in success rates between clarithromycin and amoxicillin was +1.9% (P=0.14), with a 95% CI of −0.6% to +4.5%.
3.4. Radiological Success
Radiological success rates were available for four studies (Table 4). Success rates varied between 78.2% and 94.0% in the clarithromycin group and between 79.7% and 94.0% in the amoxicillin group. For this endpoint, there was considerable heterogeneity of success-rate differences, which varied between −13.6% and +8.2% (Cochran’s Q=7.6, P=0.06), requiring a random-effects meta-analysis. The combined difference in success rates between clarithromycin and amoxicillin was 0.0% (P=1.0), with a 95% CI of −7.5% to +7.5%.
| Pathogen | n | % |
|---|---|---|
| Streptococcus pneumoniae | 237 | 31.8 |
| Haemophilusa | 213 | 28.6 |
| Staphylococcus aureus | 155 | 20.8 |
| Moraxella catarrhalisb | 65 | 8.7 |
| Enterobacteriaceae | 25 | 3.4 |
| Streptococci | 22 | 3.0 |
| Streptococcus pyogenes | 7 | 0.7 |
| Branhamella catarrhalisb | 2 | 0.3 |
| Other | 20 | 2.7 |
| Total | 746 | 100 |
| Reference | Clarithromycin | Amoxicillin | Difference Clarithromycin vs. Amoxicillin (%) | |||
|---|---|---|---|---|---|---|
| n | % | n | % | |||
| [12] | 50/55 | 90.9 | 54/61 | 88.5 | +2.4 | |
| [11] | 128/132 | 97.0 | 119/128 | 93.0 | +4.0 | |
| [13] | 115/134 | 85.8 | 110/129 | 85.3 | +0.5 | |
| [10] | 29/33 | 87.9 | 32/35 | 91.4 | –3.5 | |
| [9] | 52/57 | 91.2 | 48/57 | 84.2 | +7.0 | |
| [14] | 184/188 | 97.9 | 179/185 | 96.8 | +1.1 | |
| Combined rate difference* | +1.9 | |||||
| Cochran’s Q=2.2, P=0.82 | 95% CI –0.6 to +4.5 | |||||
| Reference | Clarithromycin | Amoxicillin | Difference Clarithromycin vs. Amoxicillin (%) | ||
|---|---|---|---|---|---|
| n | % | n | % | ||
| [12] | 43/55 | 78.2 | 56/61 | 91.8 | –13.6 |
| [11] | 116/132 | 87.9 | 102/128 | 79.7 | +8.2 |
| [10] | 30/33 | 90.9 | 31/35 | 88.6 | +2.3 |
| [14]a | 172/183 | 94.0 | 172/183 | 94.0 | 0.0 |
| Combined rate difference* | +0.5 | ||||
| Cochran’s Q=7.6, P=0.06 | 95% CI –3.5 to +4.4 | ||||
| Pathogen | Clarithromycin | Amoxicillin | ||
|---|---|---|---|---|
| n | % | n | % | |
| Streptococcus pneumoniae | 87/92 | 94.6 | 73/80 | 91.3 |
| Haemophilus | 55/62 | 88.7 | 61/64 | 95.3 |
| Staphylococcus aureusa | 49/61 | 80.3 | 51/58 | 87.9 |
| Moraxella catarrhalisb | 18/20 | 90.0 | 19/20 | 95.0 |
| Branhamella catarrhalisb | 2/2 | 100 | −/− | − |
| Streptococcus pyogenes | 2/2 | 100 | 5/5 | 100 |
| Other | 5/5 | 100 | 12/13 | 92.3 |
| Total | 218/244 | 89.3 | 221/240 | 92.1 |
3.5. Bacteriologic Cure Rates
Bacteriologic cure rates were reported for five of the six studies (Table 5). Cure rates varied between 87.1% and 94.6% with clarithromycin and between 89.8% and 98.1% with amoxicillin. The combined difference in success rates between clarithromycin and amoxicillin was −3.2% (P=0.16), with a 95% CI of −7.7% to +1.3%.
3.6. Bacteriologic Eradication
Eradication rates are shown in Table 6. On average, eradication rates were slightly higher with amoxicillin. Overall eradication rates were comparable between the two treatments (clarithromycin, 89.3%; amoxicillin, 92.1%).
3.7. Safety Data
Documented rates of adverse events varied between studies. Riffer and colleagues [14] reported a significantly higher incidence of dysgeusia with clarithromycin than amoxicillin (11% vs. 1%; P<0.001) but more vaginitis among female patients treated with amoxicillin (8% vs. 2%; P=0.028). Gastrointestinal-related adverse events were among the most often reported incidents and, in the case of Dubois et al. [11], affected a significantly greater proportion of patients treated with amoxicillin than clarithromycin (78% vs. 50%; P=0.001). Karma et al. [10] reported overall adverse event rates of 16% for clarithromycin and 26% for amoxicillin, while Gehanno et al. [13] reported corresponding rates of 14.8% and 12.2%.
4. DISCUSSION
Our data indicate broadly similar clinical, radiological and bacteriological effectiveness of clarithromycin and amoxicillin (with or without clavulanate potassium) in the setting of AMS and, to that extent, indicate that clarithromycin, while currently not routinely identified as first-line therapy for bacterial AMS, may be considered as a viable treatment option for patients who are candidates for antibiotic therapy. The high clinical success rates (85.8−97.9% for clarithromycin and 84.2−96.8% for amoxicillin) plus the determination of a non-wide CI of −0.6% to +4.5% for the comparison of clinical effectiveness illustrates the comparable efficacy of clarithromycin and amoxicillin. It should be borne in mind throughout, however, that only a small proportion of cases of AMS have bacterial causes or indications for antibiotic therapy and that use of antibiotics in this setting should be a relative niche application.
We identified six randomized comparisons of clarithro- mycin and amoxicillin in our investigations [9-14]. These recruited an initial total of 1580 patients, but exclusions from analysis predominantly attributed to non-fulfilment of inclusion criteria reduced the number of patients contributing data. Reliance on a numerically small base of data is, however, not exceptional in antibiotic research. Hernandez et al. [15] drew on limited sources in a recent meta-analysis of placebo-controlled studies of the use of antibiotics in the treatment of AMS [16]. Similarly, the 2020 European Position Paper on Rhinosinusitis and Nasal Polyps (EPOS2020), the current exhaustive English-language survey of the field, based its advice on the use of antibiotics in rhinosinusitis on three placebo-controlled, randomized (but not all of which were blinded) studies, one of which recorded no clinical benefit of antibiotic (specifically, moxifloxacin 400 mg for 5 days [17]) and another which found nasal ultrasound to be non-inferior to antibiotics (amoxicillin 500 mg t.i.d. for 10 days) for the relief of rhinosinusitis-related face pain [18]. EPOS2020 advice (graded 1a) on the use of antibiotics in bacterial rhinosinusitis and the expressed preference for beta-lactams thus rests essentially on a >20-year-old study conducted in 130 patients [17, 19]. Viewed in that context, our dataset assumes more substance. All the data used in this meta-analysis are available from peer-reviewed and publicly available reports. Our calculations are, therefore, open to independent scrutiny and corroboration.
Most of the patients enrolled in the studies we examined were adults, and the interpretation of our conclusions should be restricted accordingly.
Hernandez et al. [15] have noted that amoxicillin/ clavulanate was associated with “significantly more dropouts because of adverse effects than cephalosporins or macrolides”, and some aspects of our safety database corroborate that perception. Rechtweg et al. [20] examined the impact of amoxicillin/clavulanate and clarithromycin on quality of life in patients being treated for acute rhinosinusitis and concluded (from a sample of 20 patients) that clarithromycin may produce “a faster resolution or improvement of symptoms” vis-à-vis amoxicillin but that the two treatments are otherwise similar in their impact on quality-of-life indices [21].
Our findings for bacteriologic cure favor clarithromycin. This seeming benefit draws attention to theoretically favorable features of the pharmacokinetics and metabolism of clarithro- mycin [22, 23] and are consistent with (although not confir- matory of) evidence that clarithromycin exerts wide-ranging anti-inflammatory and immunomodulatory effects that may be pertinent in AMS [24-26], and which may not be apparent with amoxicillin [27]. A commentary on these aspects of clarithromycin, including the possible contribution of the active metabolite 14-hydroxy-clarithromycin to overall antimicrobial effect, discussion of general pharmacokinetics, sinus pene- tration, and pharmacodynamic modeling, as well as an introduction to the concept of the mutant-prevention concen- tration, has recently been published [4]. Effects on the development of a bacterial biofilm may be relevant to the clinical efficacy of all the antibiotics evaluated, but none of our selected studies was designed to explore such an effect.
No significant difference in bacteriologic cure rates was apparent in studies that used higher doses of amoxicillin, however, suggesting a possible contribution of amoxicillin dosage to that favorable comparison (data not shown). Developments in formulation science that occurred in the years after our studies had been conducted may be noteworthy in this context [28, 29]. It should also be noted (Table 5) that the numerical rates of eradication with clarithromycin varied in ways that cannot readily be explained by the difference in comparator dosages, and this again cautions against over-interpretation of these data.
Shifting patterns of antibiotic resistance are relevant when considering these data. Profiles of resistance will have altered, often substantially, in the intervening years, as evidenced, for example, by the emergence of high-level resistance against macrolides by clinical isolates of Moraxella catarrhalis in Japan [30] and China [31], and by wider shifts in antibiotic resistance around the world [32, 33]. Decisions on the use of antibiotics in AMS of presumed bacterial origin should, so far as possible, be guided by the local circumstances of pathogen resistance and by emerging trajectories of resistance develop- ment. The Canadian Choosing Wisely campaign (www. choosingwiselycanada.org) advocates restricted antibiotic usage for uncomplicated AMS as part of broader antibiotic stewardship [34]. A comprehensive discussion of the need for discipline in antibiotic prescribing is beyond the scope of this report, but we fully endorse that necessity. Schneider et al. [35] have very recently modeled the potential of point-of-care testing as one aid to prudent prescribing, with striking results. Other initiatives have met with mixed success, highlighting the need for continuing innovation [36-38].
We recognize the desirability of additional well-configured prospective controlled trials to guide antibiotic practice in AMS, but the realities of the world make such trials improbable for the foreseeable future. In their absence, exercises such as that reported here can, without being conclusive, provide useful signals to physicians seeking pharmaceutical options to treat bacterial AMS. Our analysis leads us to the view that clarithromycin may be considered a viable alternative to amoxicillin as antibiotic therapy for AMS where such therapy is warranted. Final decisions of the choice of medication will be shaped by the circumstances of individual patients.
| Item | Score |
|---|---|
| Was the study described as randomized (this includes words such as randomly, random, and randomization)? | 0/1 |
| Was the method used to generate the method of randomization described and appropriate (table of random numbers, computer-generated, etc.)? | 0/1 |
| Was the study described as double-blind? | 0/1 |
| Was the method of double-blinding described and appropriate (identical placebo, active placebo, dummy, etc.)? | 0/1 |
| Was there a description of withdrawals and dropouts? | 0/1 |
| Deduct one point if the method used to generate the sequence of randomization was described, and it was inappropriate (patients were allocated alternately or according to date of birth, hospital number, etc.) | 0/−1 |
CONSENT FOR PUBLICATION
Not applicable.
STANDARDS OF REPORTING
PRISMA guidelines and methodologies were followed in this study.
FUNDING
This research was supported by Abbott Products Operations AG, Allschwil, Switzerland.
CONFLICT OF INTEREST
Mr. Nauta is a salaried full-time employee of Abbott Healthcare Products B.V., Weesp, The Netherlands, and owns company stock.
Dr Hoban has in the past received grant support from Abbott, is currently a member of Abbott advisory boards, and also lectures for Abbott. No other conflicts to report.
ACKNOWLEDGEMENT
Abbott Products Operations AG, Allschwil, Switzerland, retained Hughes associates, Oxford, UK, to provide editorial assistance in the preparation of this report.


