All published articles of this journal are available on ScienceDirect.
Carbapenem-Sparing Antibiotic Treatment Options in Children with Extended-Spectrum β-Lactamase (ESBL) Producing Bacteria
Abstract
Introduction:
This is a single-site retrospective chart review study that sought to assess risk factors associated with antibiotic resistance and the likelihood of susceptibility to non-carbapenem antibiotics in ESBL-producing bacteria in positive cultures in pediatric patients.
Materials and methods:
ESBL-producing bacteria were present in 222 culture-positive cases. Among 177 isolates tested, 85.9% had susceptible breakpoint to piperacillin-tazobactam. Aminoglycoside susceptibility varied with low percentages among tobramycin and gentamicin (36.9% and 50.9%, respectively), but high susceptibility for amikacin (95.5%). Most isolates (77%) were susceptible to at least one oral option, but individual susceptibilities were low. Risk factors associated with ESBL acquisition were not independently associated with antibiotic resistance to amikacin, piperacillin-tazobactam, or combined oral options, sulfamethoxazole-trimethoprim, ciprofloxacin, and amoxicillin-clavulanate.
Conclusion:
When determining empiric treatment, for an isolate identified as ESBL prior to finalized susceptibilities, piperacillin-tazobactam may be a carbapenem-sparing antibiotic option to consider based on local resistance data. Oral antibiotic options may be appropriate in non-critical patients.
1. INTRODUCTION
1.1. Background
Previous studies in the literature have described risk-factors for ESBL acquisition in children and adults. Although ESBL-producing bacterial infections tend to affect adults 65 years and older at a higher rate, children are believed to be an important reservoir [1, 2]. Strategies for treatment in children using non-carbapenems are important for antimicrobial stewardship and decreasing the risk of future morbidity and mortality related to antibiotic resistance.
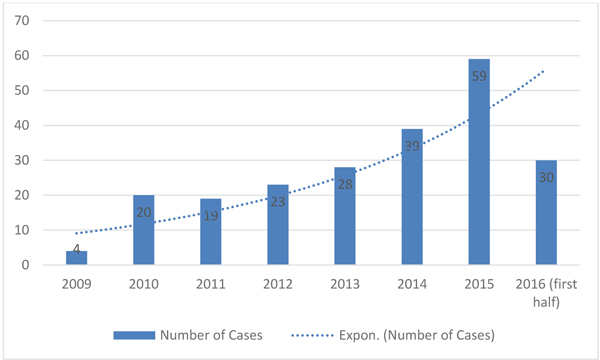
We noticed growing numbers of pediatric patients with positive cultures for ESBL-producing bacteria at our hospital over several years (Fig. 1). Extended-spectrum Beta-Lactamase (ESBL) producing bacteria have been increasing for the past two decades and emerging as a world health concern, given their resistance to available antimicrobials for treatment [1-3]. Infections involving ESBL-producing bacteria are of great concern because of their increasing antibiotic resistance and relatively higher mortality rate compared with non-ESBL-producing bacterial infections [2, 4]. ESBL-producing bacteria create plasmid-mediated enzymes (β-lactamases) that cleave the β-lactam ring making them resistant to β-lactam antibiotics such as penicillins and cephalosporins [1, 2, 5]. Furthermore, these bacteria have a genetic capacity to acquire resistance to other antibiotic classes [2]. For some cases of infection involving ESBLs, extensive co-resistance leaves carbapenems as the only choice for treatment. Carbapenems are considered the treatment of choice for these organisms overall, though many ESBLs also show susceptibility to other antibiotic options and non-carbapenem antibiotics are appropriate for some infections [2].
ESBL producing bacteria pose a challenge for the treating clinician in making choices for empiric antimicrobial coverage before culture susceptibility results are available because resistance patterns to non-carbapenems are somewhat unpredictable. Prompt appropriate antimicrobial treatment for ESBL infections is essential for successful outcomes, therefore, clinicians must use informed judgments regarding initiating carbapenems.
Infections secondary to ESBL-producing bacteria are most common in those with chronic conditions who are also at risk for multiple or prolonged hospitalizations [3, 4, 6]. We sought to find if any stable or modifiable risk factor for ESBL acquisition was associated with increased resistance to non-carbapenem antibiotics in children. Many studies have contributed to our knowledge of risk factors associated with ESBL acquisition, but few describe risk for ESBL antibiotic co-resistance in children associated with risk factors [1-7].
1.2. Goals of this Investigation
We sought to find alternatives to carbapenem use and hypothesized that a child’s personal history of chronic disease would be a useful factor to predict antibiotic resistance. Antibiotics assessed for susceptibility included ciprofloxacin, trimethoprim/ sulfamethoxazole (TMP-SMX), amoxicillin-clavulanate, gentamicin, amikacin, tobramycin, nitrofurantoin, and piperacillin-tazobactam (PTZ). Risk factors were included based on ESBL acquisition and are described in detail below.
Knowing risk factors that place children at risk of ESBL infection with highly resistant bacteria is important for judging initial treatment. Likewise, rates of antibiotic susceptibility among ESBL isolates are useful information in deciding the initial choice of empiric treatment. This information aims to guide initial treatment with non-carbapenem antimicrobials, particularly for children who are clinically stable.
2. METHODS
2.1. Study Design and Setting
This is an institutional review board approved single-center observational analytic study performed using retrospective chart review in a 248-bed freestanding pediatric hospital in Central Texas, serving the 46 surrounding counties.
2.2. Selection of Participants
Inclusion criteria consisted of all pediatric patients ages 0-18 years with a laboratory-confirmed ESBL-positive bacteria cultured from any body site between January 1, 2009, and July 1, 2016. Cases were identified by the hospital infection preventionist and included cases encountered in the emergency department, hospital-based specialty clinic, and inpatient setting. Cases of ESBL-positive isolates were included rather than individual patients to gather data specifically on risk factors and the patterns of antibiotic susceptibility among ESBL-producing bacteria.
2.3. Interventions
All isolates were reviewed for susceptibility to the following antibiotics which represent treatment options: TMP-SMX, ciprofloxacin, amoxicillin-clavulanate, gentamicin, tobramycin, amikacin, nitrofurantoin, PTZ, cefepime, and meropenem. This study relied on susceptibility testing done as standard of care, including Vitek 2 automated identification and antibiotic susceptibility tests. All isolates were tested for TMP-SMX, ciprofloxacin, gentamicin, tobramycin, amikacin, cefepime, and meropenem; nitrofurantoin was tested on urine specimens. Data for PTZ and amoxicillin-clavulanate were available in 78% and 63% of cases, respectively. Isolates were considered to have resistance if the report was intermediate or resistant.
A review of the literature was performed to identify established ESBL-related risk-factors. Each case was manually reviewed for the documented presence of nine ESBL-related risk-factors documented in the electronic hospital chart: Genitourinary (GU) abnormalities, antibiotic use in the previous three months (specifically beta-lactams, TMP-SMX, or quinolones), history of international travel or residence within the previous year, surgery within the previous year, recent or ongoing use of indwelling devices prior to current hospitalization, lifetime neonatal Intensive Care Unit (ICU) and ICU admission, history of recurrent Urinary Tract Infection (UTI), history of previous ESBL-producing bacteria, or non-GU comorbidities. ESBL risk factors were chosen based on their effect on ESBL acquisition in the literature and general availability in the pediatric chart. Other variables collected included age, sex, race/ethnicity, hospital length of stay, site of culture, treatment received for ESBL-producing bacteria, the presence of infectious diseases consult, discharge diagnosis, and ESBL-producing bacteria susceptibility profile. Due to the retrospective nature of this descriptive study, there were some data gaps in the variables assessed. The decision was made to report documented risk-factors in the chart. Cases for which a risk-factor was not addressed or not documented were coded as not having that risk-factor. Cases were manually reviewed by two study team members and a portion of that review was audited by the first reviewer.
2.4. Outcomes
The primary outcome measured in the study was resistance to non-carbapenem antibiotics. This outcome was chosen because resistance to non-carbapenem antibiotics would identify groups of children more likely to need meropenem for treatment from those who could be treated empirically with a non-carbapenem. Likewise, it would show which non-carbapenems may be most appropriate for empiric treatment. Secondly, we sought to find a relationship between ESBL-associated risk factors and resistance to non-carbapenem antibiotic options.
2.5. Analysis
Data were input into a Redcap database and analyzed using logistic regression to determine associations between ESBL-producing bacteria cultures resistant to non-carbapenems and the presence of ESBL-related risk-factors as well as differences between groups classified as children with chronic comorbidities versus children without chronic medical conditions [8]. Rstudio software was utilized for statistical testing [9].
3. RESULTS
3.1. Characteristics of Study Subjects
ESBL-producing bacteria were present in 223 cases. One case was excluded from analysis for antibiotic resistance because susceptibilities were not performed. Among the 222 cases studied, 14 isolates were repeated, individual children. The majority of the patients were female (69%) with the most common site of culture being the urinary tract (77%), followed by respiratory cultures. Table 1 shows demographics and ESBL-related risk-factors among the cases studied. Escherichia coli was the most common bacteria isolated (83%), followed by Klebsiella pneumoniae (11%). At least one known ESBL related risk-factor investigated was present in 71% of all cases. Nearly half (48%) of the patients were identified as having a history of chronic comorbidities. Half of the total cases resulted in hospital admission. Bloodstream infection was seen in 4% (9) of cases.
| Variable | Total Population (n=222) |
|---|---|
| Age (median) | 3 years |
| Sex, male | 31% (69) |
| History of Chronic Comorbidities | 48% (106) |
| History or NICU or ICU (lifetime) | 27% (61) |
| History of surgery within the past year | 26% (58) |
| History of antibiotics within the previous 3 months | 41% (91) |
| Use of Indwelling Device | 30% (66) |
| History of recurrent UTI | 26% (58) |
| International Travel or Residence within the past year | 15% (34) |
| History of ESBL positive culture | 7% (15) |
| History of non- GU comorbidity | 40% (88) |
| History of GU comorbidity | 27% (59) |
All isolates were resistant to cefepime; All isolates were susceptible to meropenem. Susceptibility to PTZ was 85.9% (177 tested). Amikacin showed susceptibility in 95.5% of cases.
Among oral therapeutic options, amoxicillin-clavulanate showed the highest susceptibility at 43.2% (testing done in 63% of cases). Among the samples tested for all 3 oral options (SMX-TMP, ciprofloxacin and amoxicillin-clavulanate), 23% of cases were resistant to all. Rates of susceptibility to each antibiotic are represented in Table 2.
| Rate of susceptibility | |
|---|---|
| Sulfamethoxazole-trimethoprim | 39.2% (87/222) |
| Ciprofloxacin | 36.4% (81/222) |
| Amoxicillin-Clavulanate | 43.2% (60/139) |
| Gentamicin | 50.9% (113/222) |
| Tobramycin | 36.9% (82/222) |
| Amikacin | 95.5% (212/222) |
| Nitrofurantoin | 89.7% (156/174) |
| Piperacillin-tazobactam | 85.9% (152/177) |
| Meropenem | 100% (222/222) |
We did not find any single risk factor or history of chronic comorbidities in patients’ history to be significantly associated with resistance to Amikacin, PTZ, or all three oral options combined. There were no isolates with resistance to all non-carbapenems and therefore, a risk factor associated with the need to meropenem treatment could not be assessed.
4. LIMITATIONS
Limitations of our study include its retrospective design which lacks a randomized matched control group. Due to the nature of this design, there were missing data due to incomplete documentation in the charts. This was handled by considering variables to be positive only if documented in the chart. Using a prospective design with complete data for each case may have affected associations between resistance to oral antibiotics and specific risk factors. Likewise, we cannot claim that data for all possible risk factors were represented in this study. For example, daycare attendance may have influenced ESBL resistance, but was not routinely asked in the chart and therefore, was not analyzed. Residence in a long-term care facility has been an important factor in the literature in adults but is very rare in our pediatric population. Additionally, there was no attempt to differentiate amongst the cases between colonization and infection with ESBL-producing bacteria
Susceptibility relied on testing done as standard of care, which may include different antibiotics based on the site of the culture. This lead to missing data among some of the antibiotics studied, including PTZ, nitrofurantoin, and amoxicillin-clavulanate
DISCUSSION AND CONCLUSION
Carbapenems are usually the preferred drug for treatment of infections caused by ESBL-producing bacteria despite emerging resistance. Choosing when to use this resource is of utmost importance in light of the reality that new antibiotics are scarce. Working to identify alternative antibiotics with likely treatment success will preserve the efficacy of carbapenems and may allow for non-carbapenem empiric therapy.
Our hypothesis that medical history may be associated with increased resistance was not supported by the data. We were unable to determine any relationship between ESBL acquisition risk factors and resistance to non-carbapenem treatment options.
Among non-carbapenem antibiotics assessed, amikacin showed the highest level of susceptibility at 95.5%. From an antimicrobial stewardship perspective, this is not the best choice because it is still a broad-spectrum antibiotic. Furthermore, amikacin carries a higher risk for nephrotoxicity and the need for level monitorization in cases of prolonged use. In our sample, PTZ shows susceptibility in 85.9% of cases. Multiple studies, largely in adult populations, looking at efficacy of PTZ caution against its’ use in high-inoculum ESBL infections, such as critically ill cases and those with bloodstream infection originating outside of the urinary tract citing higher mortality rates among this population treated with PTZ compared with a carbapenem [10]. Higher mortality has not been associated with PTZ use to treat low-inoculum cases such as those found in urine and in stable patients [10]. The research is less clear regarding bloodstream infections. It appears those with urosepsis may have a low-inoculum infection and therefore adequate coverage with PTZ [10]. The majority of the patients in our sample had positive cultures from urinary specimens (77%) and therefore, are likely well suited for treatment with PTZ as an effective alternative to meropenem.
We did not consider nitrofurantoin to be an option for statistical analysis despite high susceptibility because the limited utility of this drug outside of cystitis; in cases of UTI without suspicion of renal involvement, this may be an acceptable alternative.
We identified alternatives to carbapenems that may be utilized as empiric therapy in non-critically ill patients with risk factors of ESBL acquisition and where identification and susceptibility are not reported simultaneously.
Historically, our hospital is likely to utilize meropenem in cases of ESBL requiring hospitalization. With this data, we will seek to make changes that take it to account the severity, source of infection, and local antibiotic susceptibility patterns. For those considered to be low risk and stable, discharge on oral antibiotics with close follow up may decrease the length of stay as the majority of patients tested had a susceptibility to at least one of the 3 oral options.
We, unfortunately, did not find any modifiable or unmodifiable risk that could identify a patient as being more likely to have resistance to therapeutic options. Therefore, from a clinical standpoint, identifying established risk factors to assess the likelihood of a patient having an ESBL-caused infection may be useful, but this information cannot be used to determine the the likelihood of antimicrobial resistance/susceptibility to non-carbapenems.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
No animals/humans were used for studies that are the basis of this research.
CONSENT FOR PUBLICATION
Not applicable.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


