SYSTEMATIC REVIEW
Vitamin D and HTLV Infection: A Systematic Review
E Netto1, *, M Gomes-Neto2, C Brites3
Article Information
Identifiers and Pagination:
Year: 2019Volume: 11
First Page: 35
Last Page: 42
Publisher ID: TOIDJ-11-35
DOI: 10.2174/1874279301911010035
Article History:
Received Date: 30/12/2018Revision Received Date: 07/05/2019
Acceptance Date: 22/May/2019
Electronic publication date: 31/07/2019
Collection year: 2019

open-access license: This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International Public License (CC-BY 4.0), a copy of which is available at: https://creativecommons.org/licenses/by/4.0/legalcode. This license permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background:
Vitamin D has been associated with the pathogenesis of infectious diseases.
Objective:
To perform a systematic review on the association of vitamin D and outcomes of HTLV (Human T-cell lymphotropic virus) infection.
Methods:
We searched PubMed, LILACs, Scielo, Embase and Cochrane Library for studies addressing vitamin D and HTLV infection. We included studies published in English since 1980. Studies associated with HIV, bone metabolism and not related to HTLV- associated myelopathy/ tropical spastic paraparesis (HAM/TSP) or adult T cell leukemia/lymphoma (ATL) were excluded.
Results:
Twenty-three studies were selected and sixteen studies were included in the review (eight experimental studies, three case reports, three cases series, one cross-sectional study and one review). Fourteen studies were focused on ATL, and two on HAM/TSP. The available data show that in vitro exposure to 1,25(OH)2D inhibits proliferation of HTLV-infected lymphocytes in patients with ATL or HAM/TSP. It has been observed that hypercalcemia, the main cause of death in patients with ATL, is not associated with serum levels of 1,25(OH)2D or parathyroid hormone-related protein, but leukemia inhibitory factor/D factor seems to be an important factor for hypercalcemia pathogenesis. It was also demonstrated an association between the VDR ApaI gene polymorphism and a decreased risk of HAM/TSP in HTLV positive individuals.
Conclusion:
Despite the small number and heterogeneity of the studies, this systematic review suggests that vitamin D play a role in the pathogenesis of HTLV-associated diseases.
1. INTRODUCTION
There is an increasing interest in the non-bony functions of vitamin D [1, 2]. The cholecalciferol, 25(OH)D3, is converted into its active form in the kidney, 1,25-dihydroxycholecalciferol (1,25(OH)2D), by the enzyme 1-α-hydroxylase. Recently, it has been demonstrated that immune cells express the vitamin D receptor (VDR) [1-4]. 1,25(OH)2D is capable of suppressing T cells activity and genes involved in cell proliferation and differentiation, downregulates the release of proinflammatory cytokines such as interferon- gamma (IFN- γ), tumor necrosis factor-alpha (TNF-α), interleukin-2 (IL-2) and interleukin 12 (IL- 12), and shift T cells response from Th1 to Th2-like [5-9].
The adaptive immune response elicits the antigen-specific immune response and mediates its effect across T and B lymphocytes [10]. Vitamin D has an indirect effect on the immune activity of lymphocytes mediated by antigen-presenting cells (APC) paracrine action or promotes a direct effect mediated by VDR [11]. Some studies have observed that 1,25 (OH)2D suppresses T lymphocyte proliferation by reducing IL-2 transcription. TCD4 + (T helper - Th) lymphocytes recognize the peptides presented by APC. Depending on the type of cytokines secreted by CD4 + T lymphocytes, they can be classified into Th1, Th2, Th17, Th22 and Treg. In vitro studies demonstrated that 1,25 (OH)2 D inhibits the secretion of IFN-γ by Th1 cells [12]. Some studies have also observed that vitamin D stimulates the development of Th2 cells by increasing IL-4 synthesis, but this is a still controversial topic [13]. Treatment of T-cells in vitro with 1,25(OH)2D suppresses Th17 development [14] and inhibits IL-17 production via a post-transcriptional mechanism [15]. TCD8 + lymphocytes (cytotoxic T cells) recognize the peptides presented by nucleated cells. They have higher levels of VDR expression compared to other immune cells. Vitamin D reduces the proliferation of TCD8 + lymphocytes. Some studies have observed the action of vitamin D on the expression of cytokines secreted by TCD8 + lymphocytes, such as IL-6, IL-12, TNF-α, IL-5 and TGF-β (transforming growth factor β) [16].
Many studies have linked vitamin D levels to infectious diseases pathogenesis, including infection by, HIV [17-25]. The main risk factors related to HIV-associated hypovitaminosis D are exposure to specific antiretrovirals, chronic inflammation and immune activity associated with HIV infection [20-22]. Some studies show a faster and more severe progression of HIV infection, lower TCD4 + count, higher mortality risk and greater susceptibility to Mycobacterium tuberculosis in individuals with hypovitaminosis D [17, 21, 22, 24, 26, 27]. Insufficient production of 1,25(OH)2D in patients with HIV can induce proinflammatory cytokine generation and action [20]. High levels of proinflammatory cytokines, especially TNF-α, (which is found in persistent immune activation in HIV infection) may induce a defect in hydroxylation of 25 (OH) D3 and on its activity, by blocking the stimulatory effect of parathyroid hormone (PTH) on the action of renal 1-α-hydroxylase enzyme, which reduces conversion from the inactive form to the active form of vitamin D (1,25 (OH)2D) [23, 28-30]. Antiretrovirals (e.g. protease inhibitors and non-nucleoside reverse transcriptase inhibitors), are also risk factors for hypovitaminosis D in these patients because they interfere with vitamin D metabolism. Vitamin D deficiency did not influence the distribution of T and B lymphocytes in HIV-positive patients, but vitamin D supplementation is related to the reduction of immune activity and T-cell depletion, acting as an adjuvant to antiretroviral therapy [24].
HTLV infection causes HTLV- associated myelopathy/ tropical spastic paraparesis (HAM/TSP) and adult T cell leukemia/lymphoma (ATL), but the development of these conditions requires other associated factors. Although the risk factors associated with HTLV-1 infection outcomes are still unclear, they include high HTLV proviral load, host genetic factors (HLA haplotypes: presence of DRB1 * 0101, B * 54, absence of * 02, C * 08); viral variants (cosmopolitan HTLV-1 viral subtype); duration of infection, age and feminine gender [31-33]. The role of the immune response in HTLV pathogenesis is not fully understood, but the efficacy of the individual immune response defines the control or limitation of viral persistence and, ultimately, the outcome of HTLV-associated diseases [34-37]. HTLV can immortalize human lymphocytes, specially TCD4 + lymphocytes in ATL [38].
Due to the potential effect of vitamin D on HTLV pathogenesis, and the paucity of information in this field, we performed a systematic review on the available evidence of vitamin D effects on HTLV-associated diseases. To the best of our knowledge, this is the first review on the association between vitamin D and HTLV infection.
2. METHODOLOGY
This systematic review was performed by assessing the available data on the association between vitamin D and HTLV infection. Qualitative analysis was completed in accordance with PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines [39]
We searched PubMed, LILACs, Scielo, Embase and Cochrane Library from 1980 to February 2017. We used the keywords “VITAMIN D”, “25(OH)D”, “CALCITRIOL”, “ADULT T-CELL LYMPHOMA/LEUKEMIA” AND “HTLV” to identify related articles. We only included studies addressing vitamin D and the outcome of interest (HTLV infection), when associated with diseases such as ATL, HAM/TSP or with immune response. Only articles written in English have been included. Studies focused on HIV, bone metabolism or which had no relationship to HTLV -associated diseases were excluded. Three authors independently extracted data from the selected articles to evaluate if they fit the study questions, regarding population, outcomes, and rates of missing data.
3. RESULTS
Twenty-three studies were selected and sixteen studies were included in the review. Seven studies were excluded (one was related to HIV, two were focused on bone metabolism and four did not include HTLV-related diseases). Eight out of the sixteen included studies were experimental, five were case reports, one case series, one cross-sectional, and one review. Fourteen studies were focused on ATL, and two on HAM/TSP, as summarized in Fig. (1).
Table 1 describes the main characteristics and results of selected studies.
3.1. Effects of Vitamin D on HTLV-associated Diseases (HAM/TSP)
The literature review identified two articles reporting an association between vitamin D and HAM/TSP [40, 41]. Saito et al. conducted a study to investigate whether VDR polymorphism is associated with HAM/TSP development. They evaluated the frequency of four VDR single nucleotide polymorphisms in a group of 207 patients with HAM/TSP and 224 HTLV asymptomatic carriers. It was identified a significant association between VDR ApaI AA genotype polymorphism and a reduced risk of HAM/TSP in HTLV-infected women . However, there was no association between ApaI genotypes and HTLV proviral load, cerebrospinal fluid neopterin levels, or HTLV antibody titers in patients with HAM/TSP or in asymptomatic carriers [40]. Yamada et al. investigated the inhibitory effects of steroid hormones (dexamethasone and 1,25(OH)2D) and cyclosporin A on the synthesis of de novo DNA in four HTLV cell lines from patients with HAM/TSP. T cell lines showed high rates of expression of IL-2-receptor (IL-2R) and HLA-DR antigen. Twenty-four hours after incubation with either steroid hormones or cyclosporine A, an increase in the inhibitory effect on cellular proliferation was observed, except in the fourth lineage, which was insensitive to those drugs. Although cells presented a high level of IL-2R expression, no IL-2 production was detectable. This study suggested that the inhibition of cellular proliferation by the tested drugs in three HAM/TSP T-cell lines was not caused by the inhibition of HTLV p40 gene. They found that it was caused by the inhibition of c-myc products and other transcriptional factors, which are important proto-oncogenes that promote nuclear regulation in cellular proliferation of several T- cell leukemias [41].
3.2. Effects of Vitamin D on Adult T Cell Leukemia/Lymphoma (ATL)
Fourteen selected studies were related to ATL [42-44, 46-54, 56, 57]. Nakao et al. showed that the in vitro exposure to 1,25(OH)2D was able to inhibit the cell proliferation and de novo DNA synthesis of certain HTLV-1-positive T-cell lines. It was also observed that a potent 1,25(OH)2D analog modulates the induction of differentiation and inhibition of clonal proliferation in leukemic cells, but does not interfere in normal myeloid clonal growth [46].
Koizumi et al. evaluated the effects of 1,25(OH)2D on the regulation of mRNA involved in the proliferation of HTLV-infected T cells. Both 1,25(OH)2D and dexamethasone inhibit cellular proliferation and c-myc expression in HTLV-1-infected T-cell line. However, the rate of inhibition in c-myc mRNA expression was much higher in 1,25(OH)2D-treated cells [42].
Inoue et al. observed that 22-oxa-1,25(OH)2D, a non-calcemic analog of 1,25(OH)2D, and 1,25(OH)2D, are able to suppress both cell proliferation and PTHrP gene expression via binding to overexpressed VDR in HTLV infected T cells [43]. Elstner et al. found that another potent analog of 1,25(OH)2D, the 1,25(OH)2-20-epi-D3, has antiproliferative and differentiating effects on leukemic cells [44].
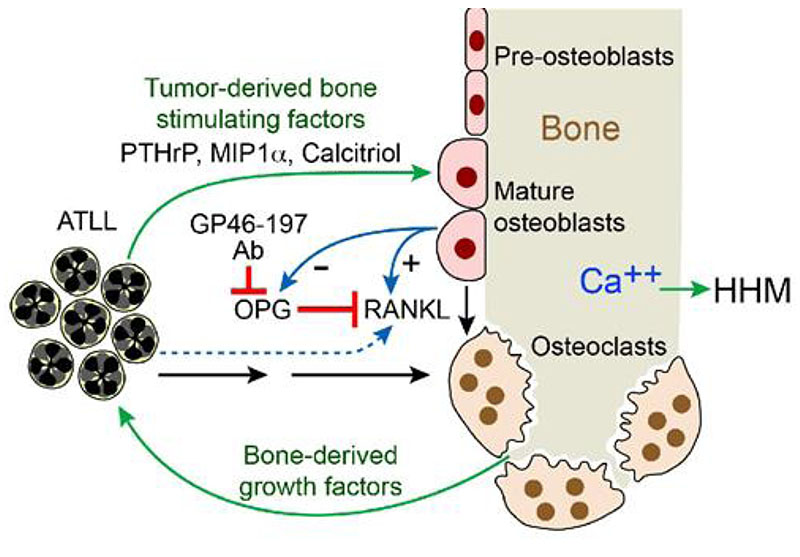
The production of 1,25(OH)2D and parathyroid hormone-related protein (PTHrP) by HTLV-infected lymphocytes was considered as a potential cause of the syndrome of humoral hypercalcemia of malignancy (HHM) frequently found in ATL patients (Fig. 2) [45]. However, this was not confirmed by several studies that failed in showing increased levels of PTHrP or 1,25(OH)2D in ATL patients [47-52]. ATL T-lymphocytes produced an osteoclast-activating-factor-like product that promoted osteoclast proliferation and hypercalcemia [52]. Dodd et al. observed that some patients with hypercalcemia and ATL had normal or suppressed levels of 1,25(OH)2D [54].
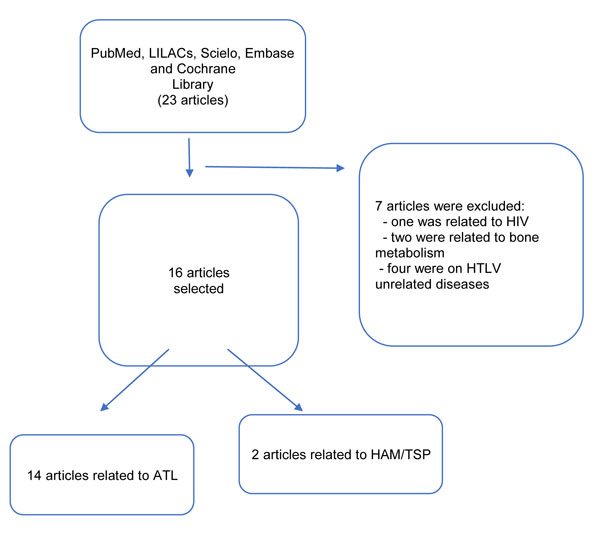 |
Fig. (1). Flowchart on the review procedures. |
Leukemia inhibitory factor/D factor (LIF) is a cytokine that affects the growth and differentiation of cell types, including hematopoietic, hepatic, adipogenic, renal, neuronal, osteoblastic and embryonic cells. Gearing et al. reported that LIF produced by osteoblastic cells stimulates bone resorption by the promotion of osteoclastic formation [55]. LIF is secreted by activated T-cells and monocytes as a part of immune response. Umemiya-Okada et al. studied the expression of LIF gene in HTLV infected T-cell lines and observed that the expression of LIF was suppressed by 1,25(OH)2D and by dexamethasone. It has been reported that HTLV infected T cells have phenotypic similarity to activated T cells, which raised the hypothesis that LIF may also contribute to the hypercalcemia and/or the immunological abnormalities of ATL [56].
Human thioredoxin (TRX) was first identified in HTLV-cell lines and has been associated with the pathophysiology of retroviral infections. TRX system has an antiapoptotic effect. Cell death induced by oxidative stress seems to be apoptosis as long as the intracellular reducing status is maintained by the TRX system [57]. Thioredoxin 2 (TRX-2) is an important regulator protein of cytochrome c release and mitochondrial apoptosis. Overexpression of TRX-2 confers an increase in mitochondrial membrane potential and resistance to etoposide-induced cell death. Vitamin D treatment upregulatesTBP-2 expression leading to growth arrest. Many tumors show downregulation of TBP-2 mRNA expression, indicating a close association between mRNA expression reduction and tumorigenesis. A growth suppressor thioredoxin binding protein-2 /vitamin D3 upregulated protein 1 (VDUP1) has its expression suppressed in HTLV-cell lines. The interaction between TBP-2/VDUP1 and TRX was documented in vitro and in vivo and apparently acts as a negative regulator of TRX. Loss of TBP-2 expression was found to be an important pathway of some HTLV cell line transformation [57].
4. DISCUSSION
This systematic review is the first one, to the best of our knowledge, to assess the role of vitamin D in HTLV infection. Despite the low number of published works on this topic, an association was observed between vitamin D and HTLV- associated diseases.
The main results of this review indicate that vitamin D can contribute to the pathogenesis of HTLV infection. It was also observed that 1,25(OH)2D inhibited, in vitro, the synthesis of de novo DNA and proliferation of HTLV infected lymphocytes [41-43]. This action could contribute to control HTLV replication. Although initial observations suggested that the synthesis of 1,25(OH)2D and PTHrP by HTLV-infected lymphocytes in patients with ATL was associated with hypercalcemia, the main cause of death in patients with ATL, subsequent studies failed to show increased levels of 1,25(OH)2D and PTHrP [38, 41]. A prospective study of subjects admitted to a hematology unit evaluated the incidence of hypercalcemia and the association with humoral mechanisms involving PTHrP, parathyroid hormone (PTH) and 1,25(OH)2D. Hypercalcemia was detected in few subjects and was caused by primary hyperparathyroidism. No evidence was found on a humoral mechanism associated with 1,25(OH)2D, but increased levels of PTHrP (similar to those seen in humoral hypercalcemia of malignancy) were detected in some patients. The relationship between elevated circulating PTHrP, and hypercalcemia during treatment, indicates a PTHrP role in the pathogenesis of hypercalcemia [58] Such association raises the hypothesis that PTHrP production by neoplastic cells in such disorders promotes a paracrine effect to induce local bone resorption. When it is released in greater amounts (generating increased circulating levels), it can promote an additional humoral-mediated effect on the development of hypercalcemia [58, 59]. Data from patients with lymphoma (including patients with AIDS-associated tumors) suggest that the deregulated synthesis of a 1,25(OH)2D-like metabolite is a frequent cause of hypercalcemia and hypercalciuria [60].
Some available evidence suggests that 1,25(OH)2D or vitamin D analogs can potently differentiate acute myeloid leukemia cells in vitro. The action of vitamin D on the inhibition of malignant cell proliferation in hematological malignancies indicates that it can be used in cancer therapy [61, 62].
VDR ApaI gene polymorphism was associated with a lower risk of developing HAM/TSP but it was not associated with the clinical course or disability degree in HAM/TSP. Six major VDR polymorphisms (CDx, A1012G, FokI, BsmI, ApaI and TaqI) have been studied in infection susceptibility to enveloped viruses [63]. In studies carried out with HIV patients, it was observed an association of VDR gene polymorphisms and HIV disease progression in injection drug users [64, 65]. In addition, another study detected an association between specific VDR gene polymorphisms and delayed disease progression rates, as well as, in resistance to HIV infection [66]. It was observed an association between rates of progression to AIDS and the VDR FokI polymorphism [52]. A meta-analysis also showed the association of FokI polymorphism with susceptibility to respiratory syncytial virus [63].
| Author | Year of publication | Study design | Number of participants | Neurological disease (HAM/TSP) | Hematologic disease (ATL) | Results |
|---|---|---|---|---|---|---|
| Fetchick et al. [48] | 1986 | Experimental (in vitro) | - | - | Yes | Production of 1,25(OH)2 D by lymphoma cells may contribute to the pathogenesis of the hypercalcemia in ATL. |
| Dodd et al. [54] | 1986 | Case series | 5 | - | Yes | Five patients with ATL had 1,25(OH)2D levels in or below the normal range and it was not associated with usual cause of hypercalcemia in such patients. |
| Nakao et al. [46] | 1987 | Experimental (in vitro) | - | - | Yes | 1,25(OH)2D inhibited the proliferation and de novo DNA synthesis of certain HTLV-positive T cell lines. |
| Kiyokawa et al. [53] | 1987 | Case series | 18 | - | Yes | 1,25(OH)2D level was low in two ATL patients with hypercalcemia. Hypercalcemia in ATL patients may be a result from the production by tumor cells of factor (s) stimulating osteoclastic bone resorption. |
| Reichel et al. [47] | 1987 | Experimental (in vitro) | - | - | Yes | HTLV-1-transformed lymphocytes can produce 1,25(OH)2D but the excess 1,25(OH)2D production was probably not associated in the pathogenesis of ATL-associated hypercalcemia. |
| Fukumoto et al. [51] | 1988 | Case series | 10 | Yes | ATL patients with hypercalcemia had low levels of 1,25(OH)2D. | |
| Koizumi et al. [42] | 1989 | Experimental (in vitro) | - | - | Yes | 1,25(OH)2D and glucocorticoid inhibited the cellular proliferation and c-myc mRNA expression in HTLV infected T-cell-line KH- 2 cells. |
| Yamada et al. [41] | 1991 | Experimental (in vitro) | - | Yes | - | Three of the four cell lines from HAM/TSP patients had their de novo DNA synthesis inhibited by steroid hormones (dexamethasone and 1,25(OH)2D) and cyclosporin A. This inhibition of cellular proliferation was not caused by the inhibition of HTLV p40 gene but by inhibition of transcriptional factors such c-myc products. |
| Umemiya-Okada et al. [56] | 1992 | Experimental (in vitro) | - | - | Yes | The expression of leukemia inhibitory factor (LIF) mRNA was suppressed by 1,25(OH)2D and dexamethasone in HTLV-1-infected T-cell lines. The expression of the LIF gene was associated in the development of hypercalcemia in ATL. |
| Johnston et al. [52] | 1992 | Case report | 1 | Yes | Elevated serum levels of 1,25(OH)2D and parathyroid hormone-related protein (PTHrP) were demonstrated in a patient with ATL hypercalcemia. | |
| Inoue et al. [43] | 1993 | Experimental (in vitro) | - | - | Yes | 22-oxa-1,25(OH)2D3 (noncalcemic analogue) and 1,25(OH)2D suppressed both cell proliferation and PTHrP gene expression through binding to the vitamin D receptor overexpressed in HTLV-1-infected T cells. |
| Elstner et al. [44] | 1994 | Experimental (in vitro) |
- | - | Yes | 1,25(OH)2-20-epi-D3, the potent 1,25(OH)2D analog, was identified with antiproliferative and differentiating effects on leukemic cells. |
| Seymour et al. [50] | 1994 | Case report | 1 | - | Yes | Hypercalcemia with normal PTHrP and vitamin D levels. |
| Peter et al. [49] | 1995 | Case report | 2 | - | Yes | Hypercalcemia with normal vitamin D levels. PTHrP seems to be the supposed factor for the hypercalcemia associated with ATL. |
| Saito et al. [40] | 2005 | Cross-sectional | 207 patients with HAM/TSP and 224 asymptomatic carriers | Yes | - | ApaI polymorphism of vitamin D receptor (VDR) was associated with the risk of HAM/TSP, although this polymorphism did not affect the proviral load of HTLV-1 in either HAM/TSP patients or asymptomatic HTLV-1 seropositive carriers. |
| Masutani H et al. [57] | 2005 | Review | - | - | Yes | Thioredoxin binding protein-2 (TBP-2)/vitamin D3 upregulated protein 1 (VDUP1) was a growth suppressor and its expression was suppressed in HTLV-1-transformed cells. |
High levels of proinflammatory cytokines (especially TNF-α, which is found in persistent immune activation in HIV infection), may induce a defect in hydroxylation and 25(OH)D3 activity by blocking the PTH stimulatory effect on renal 1-α-hydroxylase enzyme, reducing conversion from the inactive form to the active form of vitamin D (1,25 (OH)2D) [23, 30, 67]. In a preliminary study, we identified a high frequency of hypovitaminosis D in patients with HAM / TSP, and these patients had increased levels of TNF-α when compared to asymptomatic carriers of HTLV and controls (negative HTLV) (Netto, et al., submitted) [68]
Reprinted from Shu, S.; Dirksen, W.; Weibaecher, K..; Rosol, T. Mechanisms of Humoral Hypercalcemia of Malignancy in Leukemia/Lymphoma. In T Cell-Leukemia. Babusikova O., Ed.; 2011, 181-206 [45].
5. REVIEW LIMITATIONS
The results of this analysis should be interpreted with caution, due to some limitations. Firstly, there was a wide heterogeneity among studies, which did not allow us to perform a meta-analysis. In addition, most included studies were in vitro experiments with little emphasis on the clinical aspects of diseases associated with HTLV infection. Nevertheless, the available data demonstrate a potential action of vitamin D on HTLV pathogenesis and provides a rationale for a deep investigation of the relationship between vitamin D and HTLV infection.
CONCLUSION
This review demonstrates that there is an association between vitamin D and HTLV infection. However, further studies on infected HTLV patients are needed to understand the mechanisms involved in the pathogenesis of hypercalcemia in ATL and the relationship between vitamin D and HAM/TSP development.
LIST OF ABBREVIATIONS
| HIV | = Human Immunodeficiency Virus; |
| HTLV | = Human T-cell Lymphotropic Virus; |
| ATL | = Adult T cell Leukemia/Lymphoma; |
| HAM/TSP | = HTLV- Associated Myelopathy/ Tropical Spastic Paraparesis |
CONSENT FOR PUBLICATION
Not applicable.
STANDARD OF REPORTING
PRISMA Guideline and Methodology were followed.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.